Abstract
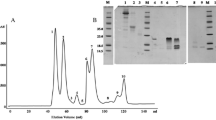
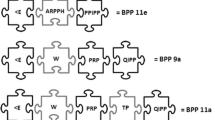
In the course of systematic investigations on low-molecular-weight compounds from the venom of Crotalidae and Viperidae, we have isolated and characterized at least three bradykinin-potentiating peptides (BPP-II, BPP-III, and BPP-V) from Bothrops neuwiedi venom by gel filtration on Sephadex G-25 M, Sephadex G-10 followed by HPLC. The peptides showed bradykinin-potentiating action on isolated guinea-pig ileum, for which the BPP-V was more active than of BPP-II, and BPP-III, rat arterial blood pressure, and a relevant angiotensin-converting enzyme (ACE) competitive inhibiting activity. The kinetic studies showed a K i of the order of 9.7 × 10−3 μM to BPP-II, 7 × 10−3 μM to BPP-III, and 3.3 ×10−3 μM to BPP-V. The amino acid sequence of the BPP-III has been determined to be pGlu-Gly-Gly-Trp-Pro-Arg-Pro-Gly-Pro-Glu-Ile-Pro-Pro, and the amino acid compositions of the BPP-II and BPP-V by amino acid analysis were 2Glu-2Gly-1Arg-4Pro-lIle and 2Glu-2Gly-lSer-3Pro-2Val-Ille, with molecular weight of 1372, 1046, and 1078, respectively.
Similar content being viewed by others
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Ferreira, L.A.F., Galle, A., Raida, M. et al. Isolation: Analysis and Properties of Three Bradykinin–Potentiating Peptides (BPP-II, BPP-III, and BPP-V) From Bothrops Neuwiedi Venom. J Protein Chem 17, 285–289 (1998). https://doi.org/10.1023/A:1022545020764
Published:
Issue Date:
DOI: https://doi.org/10.1023/A:1022545020764