Abstract
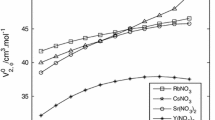
Magnesium-25 NMR measurements were carried out on aqueous and non-aqueous solutions of magnesium salts. In the former case the25Mg resonance frequency was independent of the concentration or of the counterion. In nonaqueous solvents, however, the resonance frequency was dependent on the solvent, the concentration, and on the nature of the counterion, indicating some cation-anion interactions. Measurements on Mg2+—phosphonoacetic acid mixtures in aqueous solutions gave strong indications of complexation. Only inconclusive evidence was obtained on the complexation of Mg2+ by macro-bicyclic cryptand C211 in methanol solutions, and no evidence of complexation was obtained with macrocycle 12-crown-4 in dimethylformamide solutions.
Similar content being viewed by others
References
A. I. Popov,Pure Appl. Chem. 41, 3 (1975).
F. Alder and F. C. Yu,Phys. Rev. 82, 105 (1951).
M. Ellenberger and M. Villemin,C. R. Acad. Sci. Ser. B 266 1430 (1968).
L. Simeral and G. E. Maciel,J. Phys. Chem. 80, 552 (1976).
D. H. Live and S. I. Chan,Anal. Chem. 42, 791 (1970).
H. F. Halliwell and R. C. Nyberg,Trans. Faraday Soc 59, 1126 (1963).
E. C. Ashby and R. C. Arnott,J. Organomet. Chem. 141, 1 (1968).
A. I. Popov and N. E. Skelly,J. Am. Chem. Soc. 76, 5309 (1954).
R. H. Erlich and A. I. Popov,J. Am. Chem. Soc. 93, 5620 (1971).
P.-H. Heubel and A. I. Popov, to be published.
J.-M. Lehn and J. P. Sauvage,J. Am. Chem. Soc. 97, 6700 (1975).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Heubel, PH., Popov, A.I. Magnesium-25 NMR studies of magnesium salts and complexes in nonaqueous solvents. J Solution Chem 8, 283–291 (1979). https://doi.org/10.1007/BF00650746
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00650746