Abstract
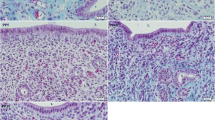
In previous studies, we have demonstrated that IGF-1 and IGF-1 receptor are expressed in rat uterus and that the expression is up-regulated by estrogen. The present study examines the expression and regulation of IGFBP-1 and IGFBP-3 in rat uterus throughout the estrous cycle. The stage of the estrous cycle in 16 mature female rats was determined by microscopic examination of daily prepared vaginal smears. Rat uteri were then used for RNA extraction. The results of the Northern blot analysis demonstrate that uterine cells express both IGFBP-1 and IGFBP-3 mRNA throughout the estrous cycle. When autoradiograms were quantitated by a densitometry, a significant reduction in expression of IGFBP-1 mRNA was found in uteri at stages of proestrous and estrous relative to that in diestrus. Although the level of IGFBP-3 mRNA varied in uteri throughout estrous cycle but this variation was not statistically significant. The lowest expression of IGFBP-1 (8.5% relative to diestrus, p<0.05, n=4) and IGFBP-3 (71% relative to diestrus) was found in the uteri prepared from rats at the stage of proestrus, while the highest expression of IGFBP-1 and IGFBP-3 was observed in the uteri obtained from rats at the stage of diestrus and metestrus, respectively. Using anti-rabbit IGFBP-1 antibody raised against an oligo-synthetic IGFBP-1 peptide, immunohistochemical staining demonstrates the presence of IGFBP-1 in the luminal and stromal glandular epithelial cells.
In summary, rat uterine cells express IGFBP-1 and IGFBP-3 mRNA and expression is regulated throughout the estrous cycle. A marked reduction in IGFBP-1 and IGFBP-3 during the proestrous stage of the estrous cycle may facilitate the biovailability of elevated IGF-1 to interact with IGF-1 receptor through a paracrine and/or autocrine mechanism.
Similar content being viewed by others
References
Kirkland JL, Gardner RM, Mukku VR, Akhtar M, Stancel GM: Hormonal control of uterine growth: the effect of hypothyroidism on estrogen stimulated cell division. Endocrinology 108: 2346–2351, 1981
Martin L, Finn CA, Trinder G: Hypertrophy and hyperplasia in the mouse uterus after estrogen treatment: an autoradiographic study. J Endocrinology 56: 133–144, 1973
Quamby VE, Korach KS: The influence of 17-b-estradiol on patterns of cell division in the uterus. Endocrinology 114: 694–702, 1984
Kirkland JL, Lapoint L, Justin E, Stancel GM: Effect of estrogen on mitosis in individual cell types of the immature rat uterus. Biol Reprod 21: 269–272, 1979
Tomocka Y, DiAugustine RP, McLachian JA: Proliferation of mouse uterine epithelial cellsin vitro. Endocrinology 118: 1011–1018, 1986
Chen L, Lindner HR, Lacet M: Mitogenic action of oestradial-17b on human myometrial and endometrial cells in long-term tissue culture. J Endocrinology 59: 87–97, 1973
Gerschenson LE, Conner E, Murai JT: Regulation of the cell cycle by diethystilbestrol and progesterone in cultured endometrial cells. Endocrinology 100: 1468–1471, 1977
Pavlik EJ, Katzenellenbogen BS: Human endometrial cells in primary tissue culture: Estrogen interactions and modulation of cell proliferation. J Clin Endocrinol Meta 47: 333–344, 1978
Stirbasku DA, Benson RH: Estrogen-inducible growth factors that may act as mediators (estromedins) of estrogen promoted tumor cell growth. In: GH Sato, R Ross (eds) Hormones and Cell Culture. Cold Spring Harbor Laboratory, Cold Spring Harbor, New York, 1979, p 477
Cooke PS, Uchima FDA, Fujii KD, Bern HA, Cunha GR: Restoration of normal morphology and estrogen responsiveness in cultured vaginal and uterine epithelia transplanted with stroma. Proc Natl Acad Sci USA 83: 2109–2113, 1986
Ghahary A, Chakrabarti S, Murphy LJ: Localization of the site of synthesis and action of insulin-like growth factor-1 in rat uterus. Mol Endocrinol 4: 191–195, 1990
Murphy LJ, Murphy LC, Friesen HG: Estrogen induces insulin-like growth factor-1 expression in the rat uterus. Mol Endocrinol 1: 445–450, 1988
Ghahary A, Murphy LJ: Uterine insulin-like growth factor-1 receptors: Regulation by estrogen and variation throughout estrous cycle. Endocrinology 125: 597–604, 1989
Hardouin S, Hossenlopp P, Segovia B, Seurin D, Portolan G, Lassarre C, Binoux M: Heterogeneity of insulin-like growth factor binding proteins and relationships between structure and affinity. Eur J Biochem 170: 121–132, 1987
Meuli C, Zapf J, Froesch ER: NSILA-carrier protein abolishes the action of nonsuppressible insulin-like activity (NSILA-S) on perfused rat heart. Daibetologia 14: 255–259, 1978
Knauer DJ, Smith GL: Inhibition of biological activity of multiplication-stimulating activity by binding to its carrier protein. Proc Natl Acad Sci USA 77: 7252–7256, 1980
Burch WM, Correa J, Shively JE, Powell DR: The 25 kilodalton insulin-like growth factor (IGF-1) binding protein inhibits both basal and IGF-1 mediated growth of chick embryo pelvic cartilagein vitro. J Clin Endocrinol Meta 70: 173–180, 1990
Murphy LJ, Seneviratne C, Ballejo G, Croze F, Kennedy TG: Identification and characterization of a rat decidual insulin-like growth factor binding protein cDNA. Mol Endocrinol 4: 329–336, 1990
Shimasaki S, Koba A, Mercado M, Shimonaka M, Ling N: Complementary DNA structure of the high molecular weight rat insulin-like growth factor binding protein (IGF-BP3) and tissue distribution of its mRNA. Biochem Biophys Res Commun 165: 907–912, 1989
Chirgwin J, Pryzbyla A, MacDonald R, Rutter W: Isolation of biological active ribonucleic acid from sources enriched for ribonuclease. Biochemistry 18: 5294–5299, 1979
delToledo SM, Murphy LJ, Hartton TH, Friesen HG: Regulation of 70-kilodalton heat shock-like messenger ribonucleic acidin vitro andin vivo by prolactin. Mol Endocrinol 1: 430–434, 1987
Luo J, Reid RE, Murphy LJ: Dexamethasone increases hepatic insulin-like growth factor binding protein-1 (IGFBP-1) mRNA and serum IGFBP-1 concentrations in the rats. Endocrinology 127: 1456–1462, 1990
Murphy LJ, Ghahary A: Uterine insulin-like growth factor-1: Regulation of expression and its role in estrogen induced uterine proliferation. Endocrine Review 11: 443–453, 1990
Krisinger J, Dann JL, Currie WD, Jeung EB, Leung PCK: Calbindin D9k mRNA is tightly regulated during the estrous cycle in the rat uterus. Mol Cell Endocrinol 86: 119–123, 1992
Fazleabas AT, Jaffe RC, Verhage HG, Waites G, Bell SC: An insulin-like growth factor binding protein in the baboon (papio anubis) endometrium: Synthesis, immunocytochemical localization and hormonal regulation. Endocrinology 124: 2321–2329, 1989
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Ghahary, A., Luo, J. & Murphy, L.J. Expression and regulation of insulin-like growth factor binding protein-1 in the rat uterus throughout estrous cycle. Mol Cell Biochem 124, 43–49 (1993). https://doi.org/10.1007/BF01096380
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01096380