Abstract
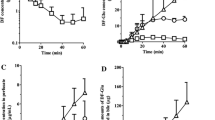
The pig has been suggested as an animal model in biomedical research because of its physiological similarity to man. Therefore, the pharmacokinetics and metabolism of diclofenac sodium (Voltaren) were studied in four Yucatan minipigs after intravenous administration of 25 and 50 mg and oral administration of 50 mg in a solution of 50 mL buffer, 50 mL water, and 200 mL water, and the results compared to historical data in man. The absolute bioavailability after oral administration of 50 mL buffer, 50 mL water, and 200 mL water solutions were 107, 97, and 109%, respectively, compared to approximately 50% in man. The total plasma clearance in minipigs was fivefold slower than in humans (57 ± 17 vs 252 ± 54 mL/hr/kg). The plasma levels of the metabolites 4′-hydroxy, 5-hydroxy, 3′-hydroxy, 4′,5-dihydroxy, and 3′-hydroxy-4′-methoxy diclofenac were considerably lower in minipigs than in man after both iv and oral administration. These results suggest slower metabolism and/or enterohepatic recirculation of the parent drug in minipigs. The volume of distribution of the central compartment was 40% less in humans than in pigs (39 vs 67 mL/kg). The terminal half-lives of the parent drug were similar in pigs (2.4 hr) and humans (1.8 hr). The rate of oral drug absorption increased in the order of 50 mL aqueous, 200 mL aqueous, and 50 mL buffered solutions (K a = 0.52±0.11, 0.59±0.13, and 1.2±0.7 hr−1, respectively). These trends are similar in man and suggest that both buffering and intake volume can affect diclofenac absorption. Possible reasons for these results include the pH-dependent solubility of this drug and the effect of volume on gastric emptying.
Similar content being viewed by others
REFERENCES
W. Reiss, H. Stierlin, P. Degan, J. W. Faigle, A. Gerardin, J. Moppert, A. Sallmann, K. Schmid, A. Schweizer, M. Sulc, W. Theobald, and J. Wagner. Pharmacokinetics and metabolism of the anti-inflammatory agent Voltaren. Scand. J. Rheumatol. Suppl. 22:17–29 (1978).
W. Reiss, H. Stierlin, J. W. Faigle, et al. The pharmacokinetics of diclofenac in animals and man. In F. J. Wagenhauser (ed.), Voltaren, a New Nonsteroidal Antirheumatic Agent (Diclofenac); Proc. VIII Congr. Rheumatol., Helsinki, 1975, pp. 19–28.
H. Stierlin, J. W. Faigle, A. Sallmann, et al. Biotransformation of diclofenac sodium (Voltaren) in animals and man I. Isolation and identification of principal metabolites. Xenobiotica 9:601–610 (1979).
H. Stierlin and J. W. Faigle. Biotransformation of diclofenac sodium (Voltaren) in animals and man. II. Quantitative determination of the unchanged drug and principal phenolic metabolites in urine and bile. Xenobiotica 9:611–621 (1980).
W. A. Cressman and D. Sumner. The dog as a quantitative model for evaluation of non-disintegrating sustained-release tablets. J. Pharm. Sci. 60:132–134 (1971).
W. Crouthamel and A. C. Sarapu. Animal Models for Oral Drug Delivery in Man: In Situ and in Vivo Approaches, Am. Pharm. Assoc., Washington, DC, 1980.
J. B. Dressman. Comparison of canine and human gastrointestinal physiology. Pharm. Res. 3:123–131 (1986).
W. G. Pond and K. A. Houpt. The Biology of the Pig, Comstock, Ithaca, NY, 1978.
M. E. Tumbleson (ed.). Swine in Biomedical Research, Plenum Press, New York, 1985.
H. Souhaili-El Amri, A. M. Batt, and G. Siest. Comparison of cytochrome P-450 content and activities in liver microsomes of seven animal species, including man. Xenobiotica 16:351–358 (1986).
C. L. Litterest, T. E. Gram, E. G. Mimnaugh, P. Leber, D. Emmerling, and R. I. Freudental. A comprehensive study of in vitro drug metabolism in several laboratory species. Drug Metab. Disp. 4:203–207 (1976).
E. Harboe, C. Larsen, M. Johansen, and H. P. Olesen. Macromolecular prodrugs. XIV. Absorption characteristics of naproxen after oral administration of a dextran T70-naproxen ester prodrug in pigs. Int. J. Pharm. 53:157–165 (1989).
J. M. Terris, T. V. Martin, and R. C. Simmonds. Metabolism unit, confinement unit and placement of indwelling catheters for use with swine. In M. E. Tumbleson (ed.), Swine in Biomedical Research, Vol. 1, Plenum Press, New York, 1985, pp. 111–121.
K. Chan, K. Vyas, and K. Wnuck. A rapid and sensitive method for the determination of diclofenac sodium in plasma by high-performance liquid chromatography. Anal. Lett. 15;1649–1663 (1982).
Statistical Consultants, Inc. PCNONLIN and NONLIN: Systems for the statistical analysis on nonlinear models. Am. Stat. 40:52 (1986).
J. C. K. Loo and S. Riegelman. New method for calculating the intrinsic absorption rate of drugs. J. Pharm. Sci. 57:918–928 (1968).
J. V. Willis, M. J. Kendall, R. M. Flinn, D. P. Thornhill, and P. G. Welling. The pharmacokinetics of diclofenac sodium following intravenous and oral administration. Eur. J. Clin. Pharmacol. 16:405–410 (1979).
W. Reiss, H. Stierlin, J. W. Faigle, V. P. Gerger, A. Gerardin, K. Schmid, M. Sulc, J. Wagner, and W. P. Theobald. Kinetik und biotransformation von diclofenac in tier und mensch. Therapiewoche 26:18 (1976).
J. O. Peggins, T. F. McMahon, W. P. Beierschmitt, and M. Weiner. Comparison of hepatic and renal metabolism of acetaminophen in male and female miniature swine. Drug Metab. Disp. 15:270–273 (1987).
C. L. Litterst, T. E. Gram, E. G. Mimnaugh, P. Leber, D. Emmerling, and R. I. Freudenthal. A comprehensive study of in vitro drug metabolism in several laboratory species. Drug Metab. Disp. 4:203–207 (1976).
J. O. Peggins, L. A. Shipley, and M. Weiner. Characterization of age-related changes in hepatic drug metabolism in miniature swine. Drug Metab. Disp. 12:379–381 (1984).
P. H. Degen, W. Dieterle, W. Schneider, W. Theobald, and U. Sinterhauf. Pharmacokinetics of diclofenac and five metabolites after single doses in healthy volunteers and after repeated doses in patients. Xenobiotica 18:1449–1455 (1988).
H. S. H. Lau, K. Chan, L. Y. Shum, S. Adair, H. Ross, H. Eyring, D. Guase, and V. John. Dose-proportionality of diclofenac sodium (Voltaren) in man. Pharm. Res. 6:S-194 (1989).
N. Vidon, A. Pfeiffer, J. Godbillon, M. Rongier, S. Gandon, J. Hirtz, J. J. Bernier, and J. P. Dubois. Evaluation of the gastric absorption and emptying of drugs under various pH conditions using a simple intubation method: Application to diclofenac. Br. J. Clin. Pharmacol. 28:121–124 (1989).
R. Menase, P. R. Hedwall, J. Kraetz, C. Pericin. L. Riesterer, A. Sallmann, R. Ziel, and R. Jagnes. Pharmacological properties of diclofenac sodium and its metabolites. Scand. J. Rheumatol. Suppl. 22:5–16 (1978).
R. L. Oberle and H. Das. Pharmacokinetics of diclofenac sodium (Voltaren) in Yucatan miniswine. Pharm. Res. 7:S-237 (1990).
M. Hossain, W. Abramowitz, B. J. Watrous, G. J. Szpunar, and J. W. Ayres. Gastrointestinal transit of non-disintegrating, non-erodible oral dosage forms in pig. Pharm. Res. 7:1099–1012 (1990).
R. L. Oberle, T. S. Chen, C. Lloyd, J. L. Barnett, C. Owyang, J. Meyer, and G. L. Amidon. The influence of interdigestive migrating myoelectric complex on the gastric emptying of liquids. Gastroenterology 99:1275–1282 (1990).
P. G. Welling. Effect of food on bioavailability of drugs. Pharm. Int. 1:14–17 (1980).
W. S. Nimmo. Gastric emptying and drug absorption. Pharm. Int. 1:221–223 (1980).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Oberle, R.L., Das, H., Wong, S.L. et al. Pharmacokinetics and Metabolism of Diclofenac Sodium in Yucatan Miniature Pigs. Pharm Res 11, 698–703 (1994). https://doi.org/10.1023/A:1018976212986
Issue Date:
DOI: https://doi.org/10.1023/A:1018976212986