Abstract
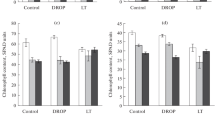
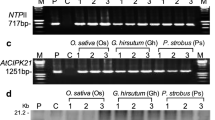
The response of plants to cold stress is not well understood at the biochemical level, although it has been studied extensively at the ecological level. To investigate whether protein phosphorylation may play an important role in cold stress, we exposed rice seedlings to low temperatures, prepared protein extracts from the leaves and incubated these in the presence of [γ-32P]ATP. The proteins were then separated by two-dimensional polyacrylamide gel electrophoresis. While several proteins were found to be phosphorylated upon cold stress one protein, pp35, which has an isoelectric point of 8.0, was more phosphorylated than the others. The pp35 protein was found to be phosphorylated when rice seedlings were incubated for 6 h at 5°C before the leaf protein extract was prepared and radioactive labeling was performed. The pp35 was, however, significantly more phosphorylated in cold-tolerant rice varieties. Antibodies were raised against purified pp35 in adult rabbits. Using this pp35 antibody, which can recognize the RuBisCO large-chain subunit (LSU), and from amino acid sequencing of pp35, we were able to identify and confirm the pp35 protein as the fragment of RuBisCO LSU (EC 4.1.1.39). Phosphorylation of the RuBisCO LSU may be important in cold tolerance.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received: 7 July 1998 / Accepted: 19 December 1998
Rights and permissions
About this article
Cite this article
Komatsu, S., Karibe, H., Hamada, T. et al. Phosphorylation upon cold stress in rice (Oryza sativa L.) seedlings. Theor Appl Genet 98, 1304–1310 (1999). https://doi.org/10.1007/s001220051196
Issue Date:
DOI: https://doi.org/10.1007/s001220051196