Summary
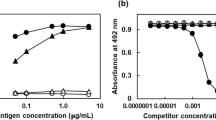
A radioimmunoassay using antibody against glucitol-lysine was developed to quantitate glycated proteins in the lens of diabetic rats. The amount of glycated protein was expressed as molar equivalents of reduced glycated hippuryl lysine (GlcRED-Hip-Lysine). Significant differences (p<0.01) were found in the amounts of glycated protein in the lenses of rats with streptozotocin-induced diabetes (3.92±0.59 nmol/mg protein, n=5), those with streptozotocin-induced diabetes treated with insulin (2.94±0.36 nmol/mg protein, n=4) and normal rats (1.23±0.22 nmol/mg protein, n=5). There was a significant correlation between the concentration of glycated protein in the lens and the HbA1c level at the end of the 12 week experiment (r=0.957, p<0.001). These results indicate that glycation of lens protein is parallel with the severity of diabetes in rats.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Mayer TK, Freedman ZR (1983) Protein glycosylation in diabetes mellitus: a review of laboratory measurements and of their clinical utility. Clin Chim Acta 127: 147–184
Kennedy L, Baynes JW (1984) Non-enzymatic glycosylation and the chronic complications of diabetes: an overview. Diabetologia 26: 93–98
Bunn HF (1981) Evaluation of glycosylated hemoglobin in diabetic patients. Diabetes 30: 613–617
Lee JH, Shin DH, Lupovitch A, Shi DX (1984) Glycosylation of lens proteins in senile and diabetes mellitus. Biochem Biophys Res Commun 123: 888–893
Mandel SS, Shin DH, Newman BL, Lee JH, Lupovitch A, Drakes GHN (1983) Glycosylation in vivo of human lens capsule (basement membrane) and diabetes mellitus. Biochem Biophys Res Commun 117: 51–56
Monnier VM, Stevens VJ, Cerami A (1979) Nonenzymatic glycosylation, sulfhydryl oxidation, and aggregation of lens proteins in experimental sugar cataracts. J Exp Med 150: 1098–1107
Chiou SH, Chylack Jr LT, Bunn HF, Kinoshita JH (1980) Role of nonenzymatic glycosylation in experimental cataract formation. Biochem Biophys Res Commun 95: 894–901
Curtiss LK, Witztum JL (1983) A novel method for generating region-specific monoclonal antibodies to modified proteins. J Clin Invest 72: 1427–1438
Hunter WH, Greenwood FA (1962) Preparation of iodine-131 labelled human growth hormone of high specific activity. Nature 194: 495–496
Lowry OH, Rosebrough NJ, Fair AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193: 265–275
Hoenders HJ, Bloemendal H (1983) Lens proteins and aging. J Gerontol 38: 278–286
Liang JN, Hershorin LL, Chylack Jr LT (1986) Non-enzymatic glycosylation in human diabetic lens crystallins. Diabetologia 29: 225–228
Rendell M, Stephen PM, Paulsen R, Valentine JL, Rasbold K, Hestorff T, Eastberg S, Shint DC (1985) An interspecies comparison of normal levels of glycosylated hemoglobin and glycosylated albumin. Comp Biochem Physiol 81: 819–822
Higgins PJ, Garlick RL, Bunn HF (1982) Glycosylated hemoglobin in human and animal red cells. Diabetes 31: 743–748
Belcher EH, Harriss EB (1959) Studies of red cell life span in the rat. J Physiol 146: 217–234
Stevens VJ, Rouzer CA, Monnier VM, Cerami A (1978) Diabetic cataract formation: potential role of glycosylation of lens crystallins. Proc Natl Acad Sci USA 75: 2918–2922
Pande A, Garner WH, Spector A (1979) Glucosylation of human lens protein and cataractogenesis. Biochem Biophys Res Commun 89: 1260–1266
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Yano, M., Hashimoto, F., Sato, M. et al. Specific radioimmunoassay of glucitol-lysine — application to lens proteins in streptozotocin-diabetic rats. Diabetologia 31, 221–224 (1988). https://doi.org/10.1007/BF00290588
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00290588