Summary
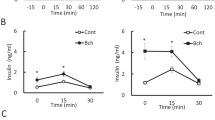
The diabetogenic action of the beta-cell toxin, alloxan, is transient when administered to mice at a dosage of 50 mg/kg. We examined whether increased cholinergic activity is involved in the compensatory mechanisms. Therefore, following administration of alloxan, methyl atropine (32 Μmol/kg) was given intraperitoneally once daily for 5 consecutive days. Methyl atropine worsened the degree of hyperglycaemia during the first week after alloxan administration. Recovery from the diabetes mellitus was observed in a substantial number of animals given alloxan without methyl atropine, whereas the risk of developing manifest diabetes was markedly enhanced by methyl atropine. At 35 days after alloxan administration, 33% of the animals, which were given alloxan alone and were diabetic after 4 days, still had diabetes. In contrast, of the animals rendered diabetic by alloxan with concomitant atropinization, 92% remained diabetic throughout the study (p=0.0145 vs alloxan alone). Glucose-stimulated insulin secretion and pancreatic insulin content were markedly reduced in animals with diabetes while being less reduced in alloxan-injected animals without diabetes. Moreover, in situ hybridization and immunocytochemistry revealed markedly decreased levels of insulin mRNA and number of insulin cells in alloxan-treated animals. With regard to insulin secretion, pancreatic insulin content, insulin mRNA and insulin cell number, the reduction was the same irrespective of whether methyl atropine had been given. Thus, 5 days of atropinization increases the incidence of diabetes following alloxan at 50 mg/kg in mice. We suggest that cholinergic activity protects insulin cells from glucotoxicity during the first week after alloxan administration and therefore, reduces the frequency of diabetes.
Similar content being viewed by others
Abbreviations
- IR:
-
Immunoreactive
References
Rerup CC (1970) Drugs producing diabetes through damage of the insulin secreting cells. Pharmacol Rev 22: 485–518
Lenzen S, Mirzaie-Petri M (1991) Inhibition of glucokinase and hexokinase from pancreatic B-cells and liver by alloxan, alloxantin, dialuric acid, and t-betylhydroperoxide. Biomed Red 12: 297–307
Dunn JS, Kirkpatrick J, McLetchie NGB, Telfer SV (1943) Necrosis of the islets of Langerhans produced experimentally. J Pathol Bact 55: 245–257
Malaisse WJ, Malaisse-Lagae F, Sener A, Pipeleers DG (1982) Determinants of the selective toxicity of alloxan on the pancreatic B-cell. Proc Natl Acad Sci USA 79: 927–930
Lenzen S, Panten U (1988) Alloxan: history and mechanism of action. Diabetologia 31: 337–342
Yamamoto H, Uchigata Y, Okamoto H (1981) Streptozotocin and alloxan induce DNA-strand breaks and poly(ADP-ribose)synthetase in pancreatic islets. Nature 294: 284–285
Ahrén B, Sundkvist G (1995) Long-term effects of alloxan in mice. Int J Pancreatol 17: 197–202
Ahrén B, Karlsson S, Lindskog S (1990) Cholinergic regulation of the endocrine pancreas. In: Aquilonius S, Gillberg PO (eds) Progress in brain research. Cholinergic neurotransmission, vol 84. Elsevier, Amsterdam, pp 209–218
Balkan B, Dunning BE (1995) Muscarinic stimulation maintains in vivo insulin secretion in response to glucose after prolonged hyperglycaemia. Am J Physiol 268:R475-R479
N'Guyen JM, Magnan C, Laury MC et al. (1994) Involvement of the autonomic nervous system in the in vivo memory to glucose of pancreatic Β cell in rats. J Clin Invest 94: 1456–1462
Weiner N (1985) Atropine, scopolamine, and related antimuscarinic drugs. In: Gilman AG, Goodman LS, Rall TW, Murad F (eds) The pharmacological basis of therapeutics, 7th edn. Macmillan, New York, pp 130–144
Mulder H, Lindh AC, Sundler F (1993) Islet amyloid polypeptide gene expression in the endocrine pancreas of the rat: a combined in situ hybridization and immunocytochemical study. Cell Tissue Res 274: 467–474
Mulder H, Ahrén B, Stridsberg M, Sundler F (1995) Non-parallelism of islet amyloid polypeptide (amylin) and insulin gene expression in rat islets following dexamethasone treatment. Diabetologia 38: 394–402
Coons AH, Leduc EM, Conolly JM (1955) Studies on antibody production. I. A method for the histochemical determination of specific antibodies and its application to a study of the hyperimmune rabbit. J Exp Med 102: 49–59
Böttcher G, Ekblad E, Ekman R, Håkanson R, Sundler F (1993) Peptide YY: a neuropeptide in the gut. Immunocytochemical and immunochemical evidence. Neuroscience 55: 281–290
Eizirik DL, Sandler S (1989) Function and metabolism of pancreatic Β-cells maintained in culture following experimentally induced damage. Pharmacol Toxicol 65: 163–168
Bonner-Weir S, Trent DF, Weir GC (1983) Partial pancreatectomy in the rat and subsequent defect in glucose-induced insulin secretion. J Clin Invest 71: 1544–1553
Rosetti L, Shulman GI, Zawalich W, DeFronzo RA (1987) Effect of chronic hyperglycaemia on in vivo insulin secretion in partially pancreatectomized rats. J Clin Invest 80: 1037–1044
Korsgren O, Jansson L, Andersson A (1989) Effects of hyperglycaemia on function of isolated mouse pancreatic islets transplanted under kidney capsule. Diabetes 38: 510–515
Ar'Rajab A, Ahrén B (1993) Long-term diabetogenic effect of streptozotocin in rats. Pancreas 8: 50–57
Korsgren O, Jansson L, Sandler S, Andersson A (1990) Hyperglycaemia-induced B cell toxicity. The fate of pancreatic islets transplanted into diabetic mice is dependent on their genetic background. J Clin Invest 86: 2161–2168
Gardemann A, Küschel GP, Jungermann K (1992) Nervous control of liver metabolism and hemodynamics. Eur J Biochem 207: 399–411
östenson CG, Grill V (1987) Evidence that hyperglycaemia increases muscarinic binding in pancreatic islets of the rat. Endocrinology 121: 1705–1710
Christensen NJ (1979) Catecholamines and diabetes mellitus. Diabetologia 16: 211–224
Bergtröm B, Lilja B, österlin S, Sundkvist G (1990) Autonomic neuropathy in non-insulin dependent (type II) diabetes mellitus. Possible influence of obesity. J Int Med 227: 57–63
Nilsson H, Bergström B, Lilja B, Juul-Möller S, Carlsson J, Sundkvist G (1995) Prospective study of autonomic nerve function in type 1 and type 2 diabetic patients: 24 hour heart rate variation and plasma motilin levels disturbed in parasympathetic neuropathy. Diabet Med 12: 1015–1021
Clauson P, Linnarsson R, GottsÄter A, Sundkvist G, Grill V (1994) Relationships between diabetes duration, metabolic control and Β-cell function in a representative population of type 2 diabetic patients in Sweden. Diabet Med 11: 794–801
Ahrén B, Taborsky Jr GJ (1986) The mechanism of vagal nerve stimulation of glucagon and insulin secretion in the dog. Endocrinology 118: 1551–1557
Karlsson S, Ahrén B (1993) Muscarinic receptor subtypes in carbachol-stimulated insulin and glucagon secretion in the mouse. J Auton Pharmacol 13: 439–446
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Ahrén, B., Sundkvist, G., Mulder, H. et al. Blockade of muscarinic transmission increases the frequency of diabetes after low-dose alloxan challenge in the mouse. Diabetologia 39, 383–390 (1996). https://doi.org/10.1007/BF00400669
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00400669