Abstract
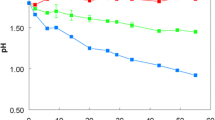
Thiobacillus ferrooxidans originally cultured on ferrous iron can adapt to grow on thiosulphate, tetrathionate, trithionate or sulphur in liquid media. In unneutralized cultures (initial pH 4–4.4) mean doubling times on thiosulphate were 8.5–13 h and on tetrathionate was about 10 h. In pH-controlled cultures, growth on thiosulphate commenced at pH values from pH 3.6–4.7, with no growth in media initially below pH 3.5 or above pH 4.8. On tetrathionate, growth commenced between pH 1.5–4.3, with no growth below pH 1.3 or above pH 4.4. Growth rate was maximal (μ = 0.043 h-1) on thiosulphate at pH 3.6 and on tetrathionate at pH 2.5 (μ = 0.05 h-1). Growth yields of about 5.9 g (210 mmoles CO2 fixed) and 9.8 g (350 mmoles CO2 fixed) dry wt per gram-molecule were obtained respectively on thiosulphate and tetrathionate. Bacteria grown on thiosulphate or tetrathionate could produce colonies on ferrous iron agar, but this ability declined on prolonged culture on sulphur compounds. All the cultures grown on various inorganic sulphur compounds were capable of re-adapting to growth on iron. This was investigated in detail. About 66–82% of iron-grown organisms readily produced colonies on tetrathionate, but only about 1 in 109 produced colonies on thiosulphate.
Similar content being viewed by others
References
Bounds, H. C., Colmer, A. R.: Comparison of kinetics of thiosulphate oxidation by three iron-sulfur oxidizers. Canad. J. Microbiol. 18, 735–740 (1972)
Brierley, J. A., Temple, K. L.: A method for preparing colloidal sulfur agar for the isolation of thiobacilli. Soil Biol. Int. News Bull. 5, 21 (1966)
Colmer, A. R.: Relation of the iron oxidizer, Thiobacillus ferrooxidans to thiosulfate. J. Bact. 83, 761–765 (1962)
Glathe, H., Ottow, J. C. G.: Ökologische und physiologische Aspekte zum Mechanismus der Eisenoxidation und Ockerbildung — eine Übersicht. Zbl. Bakt., II. Abt. 127, 750–769 (1972)
Hempfling, W. P., Vishniac, W.: Yield coefficients of Thiobacillus neapolitanus in continuous culture. J. Bact. 93, 874–878 (1967)
Hutchinson, M., Johnstone, K. I., White, D.: Taxonomy of the genus Thiobacillus: the outcome of numerical taxonomy applied to the group as a whole. J. gen. Microbiol. 57, 397–410 (1969)
Johnston, F., McAmish, L.: A study of the rates of sulfur production in acid thiosulfate solutions using S-35. J. Colloid Interf. Sci. 42, 112–119 (1973)
Kelly, D. P.: Regulation of chemoautotrophic metabolism. I. Toxicity of phenylalanine to Thiobacilli. Arch. Mikrobiol. 69, 330–342 (1969)
Kelly, D. P., Chambers, L. A., Trudinger, P. A.: Cyanolysis and spectrophotometric estimation of trithionate in mixture with thiosulphate and tetrathionate. Analyt. Chem. 41, 898–901 (1969)
Kelly, D. P., Tuovinen, O. H.: Recommendation that the names Ferrobacillus ferrooxidans Leathen and Braley and Ferrobacillus sulfooxidans Kinsel be recognized as synonyms of Thiobacillus ferrooxidans Temple and Colmer. Int. J. Syst. Bact. 22, 170–172 (1972)
Landesman, J., Duncan, D. W., Walden, C. C.: Oxidation of inorganic sulfur compounds by washed cell suspensions of Thiobacillus ferrooxidans. Canad. J. Microbiol. 12, 957–963 (1966)
Leathen, W. W.: Microbiological studies of bituminous coal mine drainage. Special summary report (Fellowship No. 326 B-6), Mellon Institute, University of Pittsburgh (1952)
Lundgren, D. G., Vestal, J. R., Tabita, F. R.: The microbiology of mine drainage pollution. In: Water pollution microbiology, R. Mitchell, Ed., pp. 69–88 New York: Wiley 1972
Margalith, P., Silver, M., Lundgren, D. G.: Sulfur oxidation by the iron bacterium Ferrobacillus ferrooxidans. J. Bact. 92, 1706–1709 (1966)
Roy, A. B., Trudinger, P. A.: The biochemistry of inorganic compounds of sulfur. Cambridge: University Press 1970
Silver, M., Lundgren, D. G.: Sulfur oxidizing enzyme of Ferrobacillus ferrooxidans (Thiobacillus ferrooxidans). Canad. J. Biochem. 46, 457–461 (1968a)
Silver, M., Lundgren, D. G.: The thiosulfate-oxidizing enzyme of Ferrobacillus ferrooxidans (Thiobacillus ferrooxidans). Canad. J. Biochem. 46, 1215–1220 (1968b)
Swaby, R. J., Vitolins, M. I.: Sulphur oxidation in Australian soils. 9th Int. Congr. Soil Sci., Trans. 4, 673–681 (1969)
Tabita, R., Silver, M., Lundgren, D. G.: The rhodanese enzyme of Ferrobacillus ferrooxidans (Thiobacillus ferrooxidans). Canad. J. Biochem. 47, 1141–1147 (1969)
Torma, A. E.: Microbiological oxidation of synthetic cobalt, nickel and zinc sulfides by Thiobacillus ferrooxidans. Rev. Canad. Biol. 30, 209–216 (1971)
Tuovinen, O. H.: Thiobacillus ferrooxidans: autotrophic growth on ferrous iron and inorganic sulphur compounds with special reference to the toxicity of metal ions and the development of resistance to uranium. Doctoral thesis. London University (1973)
Tuovinen, O. H., Kelly, D. P.: Studies on the growth of Thiobacillus ferrooxidans. I. Use of membrane filters and ferous iron agar to determine viable numbers, and comparison with 14CO2-fixation and iron oxidation as measures of growth. Arch. Mikrobiol. 88, 285–298 (1973)
Tuovinen, O. H., Kelly, D. P.: Studies on the growth of Thiobacillus ferrooxidans. II. Toxicity of uranium to growing cultures and tolerance conferred by mutation, other metal cations and EDTA. Arch. Microbiol. 95, 153–164 (1974)
Tuovinen, O. H., Niemelä, S. I., Gyllenberg, H. G.: Tolerance of Thiobacillus ferrooxidans to some metals. Antonie v. Leeuwenhoek 37, 489–496 (1971)
Unz, R. F., Lundgren, D. G.: A comparative nutritional study of three chemoautotrophic bacteria: Ferrobacillus ferrooxidans, Thiobacillus ferrooxidans and Thiobacillus thiooxidans. Soil. Sci. 92, 302–313 (1961)
Vestal, J. R., Lundgren, D. G.: The sulfite oxidase of Thiobacillus ferrooxidans (Ferrobacillus ferrooxidans). Canad. J. Biochem. 49, 1125–1130 (1971)
Author information
Authors and Affiliations
Additional information
Dedicated to the memory of a great man: Wolf Vishniac († December 10, 1973).
Rights and permissions
About this article
Cite this article
Tuovinen, O.H., Kelly, D.P. Studies on the growth of Thiobacillus ferrooxidans . Arch. Microbiol. 98, 351–364 (1974). https://doi.org/10.1007/BF00425295
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00425295