Abstract
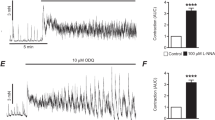
In HT29 colonie epithelial cells agonists such as carbachol (CCH) or ATP increase cytosolic Ca2+ activity ([Ca2+]i) in a biphasic manner. The first phase is caused by inositol 1,4,5-trisphophate- (Insp 3−) mediated Ca2+ release from their respective stores and the second plateau phase is mainly due to stimulated transmembraneous Ca2+ influx. The present study was undertaken to examine the effect of increased adenosine 3′,5′-cyclic monophasphate (cAMP) (forskolin 10 μmol/1 = FOR) on the Ca2+ transient in the presence of CCH (100 μmol/l). In unpaired experiments it was found that FOR induced a depolarization and reduced cytosolic Ca2+ ([Ca2+]i, measured as the fura-2 fluorescence ratio 340/380 nm) significantly. Dideoxyforskolin had no such effect. The effect of FOR was abolished when the cells were depolarized by a high-K+ solution. In further paired experiments utilizing video imaging in conjunction with whole-cell patch-clamp, [Ca2+]i was monitored separately for the patch-clamped cell and three to seven neighbouring cells. In the presence of CCH, FOR reduced [Ca2+]i uniformly from a fluorescence ratio (345/380) of 2.9 ± 0.12 to 1.8 ± 0.07 in the patch-clamped cell and its neighbours (n = 48) and depolarized the membrane voltage (V m) of the patch-clamped cells significantly and reversibly from − 54 ± 7.4 to − 27 ± 5.9 mV (n = 6). In additional experiments V m was depolarized by 15–54 mV by various increments in the bath K+ concentration. This led to corresponding reductions in [Ca2+]i. Irrespective of the cause of depolarization (high K+ or FOR) there was a significant correlation between the change in V m and change in [Ca2+]i. These data indicate that the cAMP-mediated attenuation of Ca2+ influx is caused by the depolarization produced by this second messenger.
Similar content being viewed by others
References
Allert N, Leipziger J, Greger R (1992) cAMP and Ca2+ act cooperatively on the Cl− conductance of HT29 cells. Pflügers Arch 421:403–405
Bean BP (1989) Classes of calcium channels in vertebrate cells. Annu Rev Physiol 51:367–384
Burgess GM, Bird GSJ, Obie JF, Putney JW (1991) The mechanism for synergism between phospholipase C- and adeny- lylcyclase-linked hormones in liver. J Biol Chem 266:4772–4781
Ecke D, Bleich M, Lohrmann E, Hropot M, Englert HC, Lang HJ, Warth R, Rohm W, Schwartz B, Fraser G, Greger R (1995) A chromanol type of K+ channel blocker inhibits forskolinbut not carbachol-mediated Cl− secretion in rat and rabbit colon. Cell Physiol Biochem 5:204–210
Ecke D, Bleich M, Greger R (1996) Crypt base cells show forskolin-induced Cl− secretion but no cation inward current. Pflügers Arch 431:427–434
Grynkiewicz G, Poenie M, Tsien RY (1985) A new generation of Ca2+ indicators with greatly improved fluorescence properties. J Biol Chem 260:3440–3450
Hajnoczky G, Gao E, Nomura T, Thomas AP (1993) Multiple mechanisms by which protein kinase A potentiates inositol 1,4,5-trisphosphate-induced Ca2+ mobilization in permeabilized hepatocytes. Biochem J 293:413–422
Halm DR, Rechkemmer GR, Schoumacher RA, Frizzell RA (1988) Apical membrane chloride channels in a colonie cell line activated by secretory agonists. Am J Physiol 254:C505-C511
Hamill OP, Marty A, Neher E, Sakmann B, Sigworth FJ (1981) Improved patch-clamp techniques for high-resolution current recording from cells and cell-free membrane patches. Pflügers Arch 391:85–100
Hayslett JP, Gögelein H, Kunzelmann K, Greger R (1987) Characteristics of apical chloride channels in human colon cells (HT29). Pflügers Arch 410:487–494
Horn R, Marty A (1988) Muscarinic activation of ionic currents measured by a new whole-cell recording method. J Gen Physiol 92:145–159
Kelleher DJ, Pessin JE, Ruoho AE, Johnson GL (1984) Phorbol ester induces desensitization of adenylate cyclase and phosphorylation of the β-adrenergic receptor in turkey erythrocytes. Proc Natl Acad Sci USA 81:4316–4320
Kerst G, Fischer KG, Normann C, Kramer A, Leipziger J, Greger R (1995) Ca2+ influx induced by store release and cytosolic Ca2+ chelation in HT29 colon carcinoma cells. Pflügers Arch 430:653–665
Kubitz R, Warth R, Allert N, Kunzelmann K, Greger R (1992) Small conductance chloride channels induced by cAMP, Ca2+, and hypotonicity in HT29 cells: ion selectivity, additivity, and stilbene sensitivity. Pflügers Arch 421:447–454
Kunzelmann K, Grolik M, Kubitz R, Greger R (1992) cAMP- dependent activation of small-conductance Cl− channels in HT29 colon carcinoma cells. Pflügers Arch 421:230–237
Kunzelmann K, Kubitz R, Grolik M, Warth R, Greger R (1992) Small conductance Cl− channels in HT29 cells: activation by Ca2+, hypotonie cell swelling and 9-Br-cGMP. Pflügers Arch 421:238–246
Leipziger J, Nitschke R, Greger R (1991) Transmitter-induced changes in cytosolic Ca2+ activity in HT29 cells. Cell Physiol Biochem 1:273–285
Leipziger J, Fischer KG, Greger R (1994) Voltage dependent Ca2+ influx in the epithelial cell line HT29: simultaneous use of intracellular Ca2+ measurements and nystatin perforated patchclamp. Pflügers Arch 426:427–432
Lohrmann E, Greger R (1993) Isolated perfused rabbit colon crypts: stimulation of Cl− secretion by forskolin. Pflügers Arch 425:373–380
Nitschke R, Leipziger J, Greger R (1993) Agonist induced intracellular Ca2+ transients in HT29 cells. Pflügers Arch 423: 519–526
Bleich M, Riedemann N, Warth R, Kerstan D, Leipziger J, Hör M, Van Driessche W, Greger R (1996) Ca2 mediated regulation of K+ and non-selective cation channels in colonie crypt base cells of the rat. Pflügers Arch (in press)
Schumann S, Greger R, Leipziger J (1994) Flufenamate and Gd3+ inhibit stimulated Ca2+ influx in the epithelial cell line CFPAC-1. Pflügers Arch 428:583–589
Vajanaphanich M, Schultz C, Tsien RY, Traynor-Kaplan AE, Pandol SJ, Barrett KE (1995) Cross-talk between calcium and cAMP-dependent intracellular signaling pathways. Implications for synergistic secretion in T84 colonie epithelial cells and rat pancreatic acinar cells. J Clin Invest 96:386–393
Warhurst G, Fogg KE, Higgs NB, Tonge A, Grundy J (1994) Ca2+-mobilizing agonists potentiate forskolin- and VIP-stimulated cAMP production in human colonie cell line, HT29-cl.l9A: role of [Ca2+]; and protein kinase C. Cell Calcium 15:162–174
Warth R, Riedemann N, Bleich M, van Driessche W, Busch AE, Greger R (1996) The cAMP regulated K+ conductance of rat colonie crypt base cells. Pflügers Arch (in press)
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Fischer, K.G., Leipziger, J., Rubini-Illes, P. et al. Attenuation of stimulated Ca2+ influx in colonie epithelial (HT29) cells by cAMP. Pflügers Arch. 432, 735–740 (1996). https://doi.org/10.1007/s004240050192
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s004240050192