Abstract
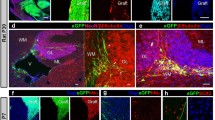
While the central nervous system (CNS) has been characterized as an immunologically privileged site, there are also several reports describing immunological reactions within the CNS. A certain degree of immunological privilege has also been ascribed to the anterior chamber of the eye. We have used the intraocular transplantation model to study immunological reactions in transplants of embryonic neural tissue. Outbred SpragueDawley rats and inbred Fisher rats were used. Pieces of rat parietal cortex or the cervical spinal cord were prepared from embryonic day 14 and implanted into the eye chambers of adult rats of the same strain. Following intraocular maturation, grafts were analysed using antibodies against: major histocompatibility complex (MHC) class I, MHC class II; rat antigens CD4, CD8, CD11b; T-cell receptor; rat antigen ED1; and glial fibrillary acidic protein. Using this set of markers for immunological reactions, transplants were scored on a blind basis. We found no significant differences in immunological scores between transplants obtained from different litters of fetuses of the outbred animals. Grafting in the outbred strain led to increased numbers of immunologically reactive cells in the grafts. This was not seen in grafts in the inbred strain. Spinal cord transplants led to a significantly higher degree of cytotoxic immunity-related cells expressing MHC class II as well as CD4-positive cells. There was a positive correlation between ED1 negativity and well-developed ramified microglia. From these results we conclude also that well-developed intraocular CNS tissue grafts do contain cellular evidence of immunological events and that different areas of the CNS may provoke different degrees of response. Reactive microglial proliferation appears to be one of the most sensitive ways to monitor the immunological condition of grafted CNS tissue.
Similar content being viewed by others
References
Barker CF, Billingham RE (1977) Immunologically privileged sites. Adv Immunol 25:1–54
Björklund H, Olson L, Seiger Å, Hoffer B (1980) Chronic lead and brain development: intraocular brain grafts as a method to reveal regional and temporal effects in the cerebral nervous system. Environ Res 224–236
Brent L (1990) Immunological privileged sites. In: Johansson B, Owman C, Widner H (eds) Pathophysiology of the blood-brain barrier. Elsevier, Amsterdam, pp 172–177
Chang Y, Albright S, Lee F (1994) Cytokines in the central nervous system: expression of macrophage colony stimulating factor and its receptor during development. J Neuroimmunol 52:9–17
Chugani DC, Kedersha NL, Rome LH (1991) Vault immunofluorescence in the brain: new insights regarding the origin of microglia. J Neurosci 11:256–268
Cserr HF, Harling-Berg CJ, Knopf PM (1992) Drainage of brain extracellular fluid into blood and deep cervical lymph and its immunological significance. Brain Pathol 2:269–276
Dallman M (1992) Graft rejection. In: Roitt IM, Delves P J (eds) Encyclopedia of immunology. Academic, London, pp 623–625
Dijkstra CD, Döpp EA, Joling P, Kraal G (1985) The heterogeneity of mononuclear phagocytes in lymphoid organs: distinct macrophage subpopulations in the rat recognized by monoclonal antibodies ED-1, ED-2 and ED-3. Immunology 54:589–599
Duan W-M, Widner H, Brundin P (1995) Temporal pattern of host responses against intrastriatal grafts of syngeneic, allogeneic or xenogeneic embryonic neuronal tissue in rats. Exp Brain Res 104:227–242
Frei K, Bodmer S, Schwerdal C, Fontana A (1986) Astrocyte-derived interleukin 3 as a growth factor for microglia cells and peritoneal macrophages. J Immunol 137:3521–3527
Fuchs HE, Bullard DE (1988) Immunology of transplantation in the central nervous system. Appl Neurophysiol 51:278–296
Gehrmann J, Banati RB, Kreutzberg GW (1993) Microglia in the immune surveillance of the brain: human microglia constitutively express HLA-DR molecules. J Neuroimmunol 48:189–198
Giacobini MMJ, Hoffer BJ, Zerbe G, Olson L (1991) Acidic and basic fibroblast growth factors segment growth of fetal brain tissue grafts. Exp Brain Res 86:73–81
Giulian D (1987) Ameboid microglia as effectors of inflammation in the central nervous system. J Neurosci Res 18:155–171
Hama T, Miyamoto M, Tsukui H, Nishio C, Hatanaka H (1989) Interleukin-6 as a neurotrophic factor for promoting the survival of cultured basal forebrain cholinergic neurons from postnatal rats. Neurosci Lett 104:304–344
Henschen A, Hoffer B, Olson L (1985) Spinal cord grafts in oculo: survival, growth, histological organization and electrophysiological characteristics. Exp Brain Res 60:38–60
Hetier E, Ayala J, Denèfle P, Bousseau A, Rouget P, Mallat M, Prochiantz A (1988) Brain macrophages synthesize interleukin-1 and interleukin-1 mRNAs in vitro. J Neurosci Res 21:391–397
Hutchinson IV (1991) Cellular mechanisms of allograft rejection. Curr Opin Immunol 3:722–728
Jordan FL, Thomas WE (1988) Brain macrophages: question of origin and interrrelationship. Brain Res Brain Res Rev 13:165–178
Kamagai M, Niijima K, Kunishita T, Nishizawa M, Ogawa M, Araki M, Ueki A, Konishi Y, Tabira T (1990) Interleukin-3 as a trophic factor for central cholinergic neurons in vitro and in vivo. Neuron 2:429–436
Kaplan HJ, Stevens TR (1975) A reconsideration of immunological privilege within the anterior chamber of the eye. Transplantation 19:302–309
Kitamura T, Miyake T, Fujita S (1984) Genesis of resting microglia in the gray matter of mouse hippocampus. J Comp Neurol 226:421–433
Koshinaga M, Whittemore SR (1995) The temporal and spatial activation of microglia in fiber tracts undergoing antegrade and retrograde degeneration following spinal cord lesion. J Neurotrauma 12:209–222
Lawrence JM, Morris RJ, Raisman G (1994) Anatomical evidence that microglia are involved in both the immune presenting and immune attack phases of intracerebral allograft rejection. Neuropathol Appl Neurobiol 20:203–205
Ling EA (1979) Transformation of monocytes into amoeboid microglia in the corpus callosum of postnatal rats, as shown by labelling monocytes by carbon particles. J Anat 128:847–58
Ling EA, Wong WC (1993) The origin and nature of ramified and amoeboid microglia: a historical review and current concepts. Glia 7:9–18
Ling EA, Kaur C, Wong WC (1991) Expression of major histocompatibility complex and leukocyte common antigens in ameboid microglia in postnatal rats. J Anat 177:117–126
Mason DW, Charlton HM, Jones A, Parry DM, Simmonds SJ (1985) Immunology of allograft rejection in mammals. In: Björklund A, Stenevi U (eds) Neuronal grafting in the mammalian CNS. Elsevier, Amsterdam, pp 91–98
Mason DW, Charlton HM, Jones AJ, Lavy CBD, Pulklavec M, Simmonds SJ (1986) The fate of allogeneic and xenogeneic neuronal tissue transplanted into the third ventricle of rodents. Neuroscience 19:685–694
Medawar PB (1958) The homograft reaction. Proc R Soc Lond [Biol] 149:144–176
Nicholas MK, Arnason BGW (1990) A role for CD8+ T lymphocytes late in the rejection of intraventricular fetal neocortical fragment allografts in the mouse. In: Johansson B, Owman C, Widner H (eds) Pathophysiology of the blood-brain barrier. Elsevier, Amsterdam, pp 573–586
Niederkorn JY (1987) Exogenous recombinant interleukin-2 abrogates anterior chamber-associated immune deviation. Transplantation 43:523–528
Niederkorn JY, Streilein JW (1982) Analysis of antibody production induced by allogeneic tumor cells inoculated into the anterior chamber of the eye. Transplantation 33:573–577
Niederkorn JY, Streilein JW (1983) Alloantigens placed into the anterior chamber of the eye induce specific suppression of delayed-type hypersensitivity by normal cytotoxic T lymphocytes and helper T lymphocyte responses. J Immunol 131: 2670–2674
Niederkorn JY, Waltenbaugh C, Streilein JW (1984) Abrogation of anterior chamber-induced suppression of delayed-types hypersensitivity responses by monoclonal anti-J antibodies. Transplantation 37:623–625
Nieto-Sampedro M, Chandy KG (1987) Interleukin-2 like activity in injured rat brain. Neurochem Res 12:723–727
Olson L (1988) Grafting in the mammalian central nervous system: basic science with clinical promise. In: Magistretti P (ed) Discussions in neurosciences. FESN, Geneva, pp 1–73
Olson L, Seiger A (1972) Brain tissue transplanted to the anterior chamber of the eye. 1. Fluorescence histochemistry of immature catecholamine and 5-hydroxytryptamine neurons reinnervating the rat iris. Z Zellforsch Mikrosk Anat 135:175–94
Olson L, Seiger Å, Strömberg I (1983) Intraocular transplantation in rodents. A detailed account of the procedure and examples of its use in neurobiology with special reference to brain tissue grafting. In: Fedoroff S (ed) Advances in cellular neurobiology, vol 4. Academic, New York
Parkman R (1992) Graft-versus-host reaction. In: Roitt IM, Delves PJ (eds) Encyclopedia of immunology. Academic, London, pp 626–627
Perry VH, Gordon S (1988) Macrophage and microglia in the nervous system. Trends Neurosci 11:273–277
Perry VH, Hume DA, Gordon S (1985) Immunohistochemical localization of macrophages and microglia in the adult and developing mouse brain. Neuroscience 15:313–326
Poltorak M, Isono M, Kulaga H, Adams AJ, Freed WJ (1992) Mechanism of immune response to intracerebral allografts in the model of allograft rejection induced by systemic immunization with donor tissue. J Neurol Transplant Plast 3:178–179
Shimizu Y, Newman W, Tanaka Y, Shaw S (1992) Lymphocyte interactions with endothelial cells. Immunol Today 13:106–112
Shinoda M, Hudson JL, Strömberg I, Hoffer BJ, Moorehead JW, Olson L (1995) Allogeneic grafts of fetal dopamine neurons: immunological reactions following two types of immunizations. Brain Res 680:180–195
Sloan DJ, Baker BJ, Puklavec M, Charlton HM (1990) The effect of site of transplantation and histocompatibility differences on the survival of neural tissue transplanted to the CNS of defined inbred rat strains. Prog Brain Res 82:141–152
Sloan DJ, Wood MJ, Charlton HM (1991) The immune responses to intracerebral neural grafts. Trends Neurosci 14:341–346
Sminia T, De Groot CJA, Dijkstra CD, Koetsier JC, Polman CH (1987) Macrophage in the central nervous system of the rat. Immunobiology 174:43–50
Stichel CC, Muller HW (1994) Extensive and long-lasting changes of glial cells following transection of the postcommissural fornix in the adult rat. Glia 10:89–100
Streilein JW, Niederkorn JY (1985) Characterization of the supressor cell(s) responsible for anterior chamber-associated immune deviation (ACAID) induced in BALB/c mice by p815 cells. J Immunol 134:1381–1387
Watschinger B, Gallon L, Carpenter C B, Sayegh M H (1994) Mechanism of allo-recognition. Recognition by in vivo-primed T cell of specific major histocompatibility complex polymorphisms presented as peptides by responder antigen-presenting cells. Transplantation 57:572–57694
Weller RO, Kida S, Zhang ET (1992) Pathways of fluid drainage from the brain — morphological aspects and immunological significance in rat and man. Brain Pathol 2:277–284
Welsh K, Male D (1989) Transplantation and rejection. In: Roitt I, Brostoff J, Male D (eds) Immunology, 2nd edn. Churchill Livingstone, London, pp 24.1–24.10
Wilbanks GA, Mammolenti M, Streilein JW (1992) Studies on the induction of anterior chamber-associated immune deviation (ACAID). III. Induction of AICAD depends upon intraocular transforming factor-β. Eur J Immunol 22:165–173
Yamada S, Depasquale M, Patlak CS, Cserr HF (1991) Albumin outflow into deep cervical lymph from different regions of rabbit brain. Am J Physiol 261:1197–1204
Zhou H, Lund RD (1993) Effects of the age of donor or host tissue on astrocyte migration from intracerebral xenografts of corpus calosum. Exp Neurol 122:155–164
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Shinoda, M., Giacobini, M., Schmidt-Kastner, R. et al. Differential immune responses to fetal intracameral spinal cord and cortex cerebri grafts. Exp Brain Res 110, 223–234 (1996). https://doi.org/10.1007/BF00228554
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00228554