Summary
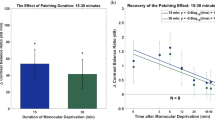
The relative efficacy of distributed versus massed reverse occlusion therapy in promoting recovery from the anatomical and physiological effects of monocular deprivation was studied in two experiments performed on kittens raised with one eye occluded from eye-opening until 5 weeks of age. The first experiment explored the effects of different periods (ranging from 0.5 to 4 h) of reverse occlusion imposed daily for 20 days. The second, involving a fixed period of reverse occlusion (20 h), examined recovery with respect to the distribution of that period over a varied number of brief daily sessions. Recovery was assessed in terms of changes in cortical ocular dominance and lateral geniculate cell morphology. Although recovery of both cortical ocular dominance and geniculate cell morphology showed the same overall progression with increasing periods of reverse occlusion, changes were apparent in the lateral geniculate nucleus before changes were evident in cortical ocular dominance. A given period of reverse occlusion was found to be far more effective in promoting recovery when distributed over a number of different exposure sessions than when massed together in just one or two sessions. The data suggest that there is a maximal rate of cortical recovery which is achieved with surprisingly brief daily periods of forced visual exposure of the initially deprived eye.
Similar content being viewed by others
References
Blakemore C, Van Sluyters RC (1974) Reversal of the physiological effects of monocular deprivation in kittens: further evidence for a sensitive period. J Physiol (Lond) 237: 195–216
Cleland BG, Mitchell DE, Crewther SG, Crewther DP (1980) Visual resolution of retinal ganglion cells in monocularly-deprived cats. Brain Res 192: 261–266
Crewther DP, Crewther SG, Mitchell DE (1981) The efficacy of brief periods of reverse occlusion in promoting recovery from the physiological effects of monocular deprivation in kittens. Invest Ophthalmol Vis Sci 21: 357–362
Crewther DP, Crewther SG, Pettigrew JD (1978) A role for extraocular afferents in post-critical period reversal of monocular deprivation. J Physiol (Lond) 282: 181–195
Derrington AM, Hawken MJ (1981) Spatial and temporal properties of cat geniculate neurones after prolonged deprivation. J Physiol (Lond) 314: 107–120
Dews PB, Wiesel TN (1970) Consequences of monocular deprivation on visual behaviour in kittens. J Physiol (Lond) 206: 437–455
Dürsteler MR, Garey LJ, Movshon JA (1976) Reversal of the morphological effects of monocular deprivation in the kitten's lateral geniculate nucleus. J Physiol (Lond) 261: 189–211
Freeman RD, Olson CR (1979) Is there a “consolidation” effect for monocular deprivation? Nature (Lond) 282: 404–406
Ganz L, Fitch M (1968) The effect of visual deprivation on perceptual behaviour. Exp Neurol 22: 639–660
Garey LJ, Dürsteler MR (1975) Reversal of deprivation effects in the lateral geniculate nucleus of the cat. Neurosci Lett 1: 19–23
Garey LJ, Fisken RA, Powell TPS (1973) Effects of experimental deafferentation on cells in the lateral geniculate nucleus of the cat. Brain Res 52: 363–369
Giffin F, Mitchell DE (1978) The rate of recovery of vision after early monocular deprivation in kittens. J Physiol (Lond) 274: 511–537
Guillery RW (1972) Binocular competition in the control of geniculate cell growth. J Comp Neurol 144: 117–127
Guillery RW, Stelzner DJ (1970) The differential effects of unilateral lid closure upon the monocular and binocular segments of the dorsal lateral geniculate nucleus in the cat. J Comp Neurol 139: 411–422
Hubel DH, Wiesel TN (1962) Receptive fields, binocular interaction and functional architecture in the cat's visual cortex. J Physiol (Lond) 160: 106–154
Hubel DH, Wiesel TN (1970) The period of susceptibility to the physiological effects of unilateral eye closure in kittens. J Physiol (Lond) 206: 419–436
Hubel DH, Wiesel TN, LeVay S (1975) Functional architecture in area 17 in normal and monocularly deprived macaque monkeys. Cold Spring Harbor Symp Quant Biol 40: 204–259
Kalil RE (1980) A quantitative study of the effects of monocular enucleation and deprivation on cell growth in the dorsal lateral geniculate nucleus of the cat. J Comp Neurol 189: 483–524
Kratz KE, Mangel SC, Lehmkuhle S, Sherman SM (1979) Retinal X- and Y- cells in monocularly lid-sutured cats: normality of spatial and temporal properties. Brain Res 172: 545–551
Kupfer C, Palmer P (1964) Lateral geniculate nucleus: histological and cytochemical changes following afferent denervation and visual deprivation. Exp Neurol 9: 400–409
Lehmkuhle S, Kratz KE, Mangel SC, Sherman SM (1978) Effects of early monocular lid suture on spatial and temporal sensitivity of neurones in dorsal lateral geniculate nucleus of the cat. J Neurophysiol 43: 542–556
Levick WR (1972) Another tungsten microelectrode. Med Electron Biol 10: 510–515
Maffei L, Fiorentini A (1976) Monocular deprivation in kittens impairs the spatial resolution of geniculate neurones. Nature (Lond) 264: 754–755
Mitchell DE, Cynader M, Movshon JA (1977) Recovery from the effects of monocular deprivation in kittens. J Comp Neurol 176: 53–64
Movshon JA (1976a) Reversal of the physiological effects of monocular deprivation in the kitten's visual cortex. J Physiol (Lond) 261: 125–174
Movshon JA (1976b) Reversal of the behavioural effects of monocular deprivation in the kitten. J Physiol (Lond) 261: 175–187
Movshon JA, Blakemore C (1974) Functional reinnervation in kitten visual cortex. Nature (Lond) 251: 504–505
Mower CD, Christen WG (1982) Effects of early monocular deprivation on the acuity of lateral geniculate neurons in the cat. Dev Brain Res 3: 475–480
Peck CK, Blakemore C (1975) Modification of single neurons in the kitten's visual cortex after brief period of monocular visual experience. Exp Brain Res 22: 57–68
Pettigrew JD, Garey LJ (1974) Selective modification of single neuron properties in the visual cortex of kittens. Brain Res 66: 160–164
Ramachandran VS, Ary M (1981) Evidence for a “consolidation” effect during changes in the ocular dominance of cortical neurons in kittens. Neurosci Abst 7: 220–221
Sanderson KJ (1971) The projection of the visual field of the lateral geniculate and medial interlaminar nuclei in the cat. J Comp Neurol 143: 101–118
Shapley RM, So YT (1980) Is there an effect of monocular deprivation on the proportions of X and Y cells in the cat's lateral geniculate nucleus? Exp Brain Res 39: 41–48
Shatz CJ, Lindstrom SH, Wiesel TN (1977) The distribution of afferents representing the right and left eyes in the cat's visual cortex. Brain Res 131: 103–116
Shatz CJ, Stryker MP (1978) Ocular dominance in layer IV of the cat's visual cortex and the effects of monocular deprivation. J Physiol (Lond) 282: 267–283
Sherman SM, Stone J (1973) Physiological normality of the retina in visually deprived cats. Brain Res 60: 224–230
Sireteanu R, Hoffman KP (1979) Relative frequency and visual resolution of X- and Y- cells in the LGN of normal and monocularly deprived cats: interlaminar differences. Exp Brain Res 34: 591–603
Wiesel TN, Hubel DH (1963a) Single cell responses in striate cortex of kittens deprived of vision in one eye. J Neurophysiol 26: 1003–1017
Wiesel TN, Hubel DH (1963b) Effects of visual deprivation on morphology and physiology of cells in the cat's lateral geniculate body. J Neurophysiol 26: 978–993
Author information
Authors and Affiliations
Additional information
Supported by a grant (78/3589) to D.E.M. and S.G.C. from the National Health and Medical Research Council of Australia
Rights and permissions
About this article
Cite this article
Crewther, S.G., Crewther, D.P. & Mitchell, D.E. The effects of short-term occlusion therapy on reversal of the anatomical and physiological effects of monocular deprivation in the lateral geniculate nucleus and visual cortex of kittens. Exp Brain Res 51, 206–216 (1983). https://doi.org/10.1007/BF00237196
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00237196