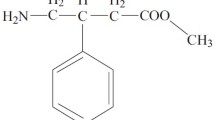
Summary
The bioavailability of quinidine in two sustained release preparations A and B has been compared in three females and three males with i.v. administration of quinidine. The initial rate of oral absorption did not differ between the two drug preparations; the peak concentration was observed after 4 h both for A and B, but was significantly higher after B. A slower decrease in plasma concentration was observed after preparation A than B. Absolute bioavailability did not differ significantly between A (median value 78.4%) and B (median 87.1%). Drug absorption in vivo was in good agreement with the results of in vitro dissolution tests on both preparations. The slower decrease in plasma concentration found for the new sustained release form of quinidine should be of clinical advantage.
Similar content being viewed by others
References
Selzer, A., Wray, H. W.: Paroxysmal ventricular fibrillation occurring during therapy of chronic atrial arrhythmias. Circulation30, 17–26 (1964)
Storstein, O.: Syncope and sudden death as a side-effect of treatment with anti-arrhythmic drugs. In: Symposium on cardiac arrhythmias s. E. Sandöe, W. Flensted-Jense, K. H. Olesen (eds.), pp. 505–514, Södertälje: AB Astra 1970
Reynolds, E. W., Vander Ark, C. R.: Quinidine syncope and the delayed repolarization syndromes. Mod. Concepts Cardiovasc. Dis.45, 117–122 (1976)
Bjerkelund, C., Skåland, K.: Kinidin som årsak til paroxystisk ventrikkelfimmer. Nord. Med.77, 76–81 (1967)
Solokow, M., Perloff, D. B.: The clinical pharmacology and use of quinidine in heart disease. Prog. Cardiovasc. Dis.3, 316–330 (1961)
Lindseth Ditlefsen, E. M.: Concentrations of quinidine in blood following delayed absorption tables. Acta Med. Scand.149, 49–53 (1954)
Cramer, G., Varnauskas, E., Werkö, L.: A new quinidine preparation with sustained release. Acta Med. Scand.173, 511–519 (1963)
Cramer, G., Isaksson, B.: Quantitative determination of quinidine in plasma. Scand. J. Clin. Lab. Invest.15, 553–556 (1963)
Koch-Weser, J.: Antiarrhythmic prophylaxis in ambulatory patients with coronary heart disease. Arch. Intern. Med.129, 763–772 (1972)
Greenblatt, D. J., Pfeifer, H. J., Ochs, H. R., Franke, K., Mac Laughlin, D. S., Smith, T. W., Koch-Weser, J.: Pharmacokinetics of quinidine in humans after intravenous, intramuscular and oral administration. J. Pharmacol. Exp. Ther.202, 365–378 (1977)
Edwards, I. R., Hancock, B. W.: Correlation between plasma quinidine and cardiac effect. Br. J. Clin. Pharmacol.1, 455–459 (1974)
Conrad, K. A., Molk, B. L., Chidsey, C. A.: Pharmacokinetic studies of quinidine in patients with arrhythmias. Circulation55, 1–7 (1977)
Crouthamel, W. G.: The effect of congestive heart failure on quinidine pharmacokinetics. Am. Heart J.90, 335–339 (1975)
Ueda, C. T., Dzindzio, B. S.: Quinidine kinetics in congestive heart failure. Clin. Pharmacol. Ther.23, 158–164 (1978)
Gibaldi, M., Perrier, D.: Pharmacokinetics. New York: Marcel Dekker 1975
Johansson, R., Regardh, C. G., Sjögren, J.: Absorption of alprenolol in man from tablets with different rates of release. Acta Pharm. Suec.8, 59–70 (1971)
Graffner, C., Johansson, G., Sjögren, J.: Comparative absorption and slow release tablets. Acta Pharm. Suec.11, 125–132 (1974)
Mahon, W. A., Mayersohn, M., Inaba, T.: Disposition kinetics of two oral forms of quinidine. Clin. Pharmacol. Ther.19, 566–575 (1976)
Ueda, C. T., Williamson, B. J., Dzindzio, B. S.: Absolute quinidine bioavailability. Clin. Pharmacol. Ther.20, 260–265 (1976)
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Amlie, J.P., Storstein, L., Olsson, B. et al. Absolute bioavailability of quinidine in two sustained release preparations. Eur J Clin Pharmacol 16, 45–48 (1979). https://doi.org/10.1007/BF00644965
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00644965