Summary
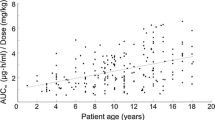
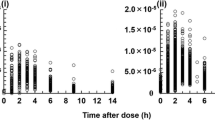
A pharmacokinetic study of bredinin, a new immunosupressive agent, was carried out in 28 renal transplant patients. Serum bredinin concentration-time curves were analyzed using a one-compartment open model with a first order absorption process. The peak serum bredinin level appeared 2.4 h after oral administration of bredinin 50–200 mg. The calculated mean peak serum level was 0.852 µg/ml/mg/kg, when the dose was adjusted to the body weight of the patient. In the dosage range used of 0.85–4.46 mg/kg, a linear relationship was observed between the dose and the peak serum bredinin level. The elimination rate of bredinin from serum was dependent on kidney function, and the elimination rate constant was well correlated with the endogenous creatinine clearance. No circadian rhythm was apparent in the elimination rate constant. The absorption rate of bredinin from the gastrointestinal (GI) tract was affected by GI diseases. The need for dosage adjustment based on the renal function of the transplant patient is suggested.
Similar content being viewed by others
References
Dettli L (1977) Elimination kinetics and dosage adjustment of drugs in patients with kidney disease. Gustav Fischer, Stuttgart New York
Gibaldi M, Perrier D (1975) Pharmacokinetics. Marcel Dekker, New York, pp 33–43
Hayashi M, Hirano T, Yaso M, Mizuno K, Ueda T (1975) Studies on bredinin. III Chemical synthesis of bredinin (A novel imidazole nucleoside). Chem Pharm Bull 23: 245–246
Inou T, Kusaba R, Takahashi I, Sugimoto H, Kuzuhara K, Yamada Y, Yamauchi J, Otsubo O (1980) Clinical trial of bredinin in renal transplantation. Transplant Proc 12: 526–528
Kaplowitz N, Kohlenkamp (1978) Inhibition of hepatic metabolism of azathioprine in vivo. Gastroenterology 74: 90–92
Mizuno K, Tsujino M, Takada M, Hayashi M, Atsumi K, Asano K, Matsuda T (1974) Studies on bredinin. I Isolation, characterization and biological properties. J Antibiot (Tokyo) 27: 775–782
Murase J, Mizuno K, Kawai K, Nishiumi S, Kobayashi Y, Hayashi M, Morino T, Suzuki T, Baba S (1978) Absorption, distribution, metabolism and excretion of bredinin in rats. Appl Pharmacol (Tokyo) 15: 829–835
Nakagawa T, Koyanagi Y, Togawa H (1979) Users manual for SALS. Computer Center of the University of Tokyo, Tokyo, Japan
Ozaki A, Iwasaki Y, Miyazima T (1980) Withdrawal of azathioprine after renal transplantation. Transplant Proc 12: 513–514
Sagawa S, Ishibashi M, Arima M, Usami M, Nagafune K, Sonoda T, Iguchi M, Kurita T, Tsujino M, Mizuno K, Murase J (1978) Immunosupressive effect of bredinin on canine kidney allotransplant survival. Transplantation (Tokyo) 13: 271–276
Sakaguchi K, Tsujino M, Yoshizawa M, Mizuno K, Hayano K (1975) Action of bredinin on mammalian cells. Cancer Res 35: 1643–1648
Sheriff MHR, Yahya T, Lee HA (1978) Is azathioprine necessary in renal transplantation. Lancet 1: 118–120
Takada K, Nakae H, Asada S, Ichikawa Y, Fukunishi T, Sonoda T (1981) Rapid method for the high-performance liquid chromatographic determination of bredinin in human serum. J Chromatogr 222: 156–159
Uchida H, Yokota K, Akiyama N, Masaki Y, Aso K, Okubo M, Okudaira M, Kato M, Kashiwagi N (1979) Effectiveness of a new drug, bredinin, on canine kidney allotransplantation survival. Transplant Proc 11: 865–870
Yoshioka H, Nakatsu K, Hayashi M, Mizuno K (1975) Studies on bredinin. II The molecular structure of bredinin. Tetrahedron Lett 46: 4031–4034
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Takada, K., Asada, S., Ichikawa, Y. et al. Pharmacokinetics of bredinin in renal transplant patients. Eur J Clin Pharmacol 24, 457–461 (1983). https://doi.org/10.1007/BF00609886
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00609886