Summary
The pharmacokinetics and effects on blood pressure and heart rate of nisoldipine were studied in 8 patients with cirrhosis and in 8 age-matched healthy controls. On separate occasions each subject received nisoldipine by i.v. infusion (0.37 mg in 40 min) and as a tablet (5 mg for patients and 20 mg for control subjects).
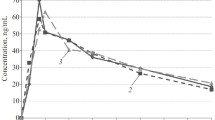
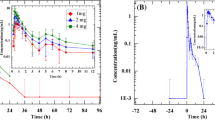
After i.v. nisoldipine, the elimination half-life was 9.7 h in control subjects and 16.6 h in the cirrhotics. The volume of distribution was 4.1 l/kg and 6.4 l/kg and the total systemic clearance was 847 ml/min and 494 ml/min, respectively. On oral nisoldipine, systemic availability was fourfold higher in patients with cirrhosis: 14.7% versus 3.7%.
After i.v. administration of nisoldipine there was a brief decrease in systolic and diastolic blood pressure in both groups, whereas the heart rate increased. After 4 h a second effect peak appeared in the control subjects. After oral nisoldipine similar effect-time profiles were found, but effects lasted longer than after i.v. administration.
Comparison of the maximal total plasma concentration of nisoldipine and the maximal effect in the two groups revealed that sensitivity to nisoldipine was not different in patients with cirrhosis.
A reduction in the dose of nisoldipine is recommended when cirrhotics require oral nisoldipine in therapeutic practice.
Similar content being viewed by others
References
Pasanini F, Meredith PA, Reid JL (1985) The pharmacodynamics and pharmacokinetics of a new calcium antagonist nisoldipine in normotensive and hypertensive subjects. Eur J Clin Pharmacol 29: 21–24
Vogt A, Neuhaus KL, Kreuzer H (1980) Hemodynamic effects of the new vasodilator drug Bay k 5552 in man. Arzneimittelforsch 30: 2162–2164
Kimchi A, Ellrodt AG, Charuzi Y, Shell W, Murata GH (1985) Salutary hemodynamic and sustained clinical benificial effects of nisoldipine, a new calcium channel blocker, in patients with recurrent ischemia and severe heart failure. Am Heart J 110: 496–498
Lam J, Chaitman BR, Crean P, Blum R, Waters DD (1985) A dose-ranging, placebo-controlled, double-blind trial of nisoldipine in effort angina: duration and extent of antianginal effects. J Am Coll Cardiol 6: 447–452
Kazda S, Garthoff B, Meyer H, Schlossmann K, Stoepel K, Towart R, Vater W, Wehinger E (1980) Pharmacology of a new calcium channel antagonistic compound, isobutyl methyl 1,4-dihydro-2,6-dimethyl-4-(2-nitrophenyl)-3,5-pyridine-dicarboxylate (Nisoldipine, Bay k 5552). Arzneimittelforsch 30: 2144–2162
Debbas NMG, Jackson SHD, Turner P (1986) A comparison of the haemodynamic effects of nifedipine, nisoldipine and nitrendipine in man. Eur J Clin Pharmacol 30: 393–397
Harten J van, Brummelen P van, Lodewijks MThM, Danhof M, Breimer DD (1988) Pharmacokinetics and hemodynamic effects of nisoldipine and its interaction with cimetidine. Clin Pharmacol Ther (in press)
Ahr G, Wingender W, Kuhlmann J (1987) Pharmacokinetics of nisoldipine. In: (eds) Hugenholtz PG, Meyer J Nisoldipine 1987. Springer, Berlin Heidelberg, pp. 59–66
Kleinbloesem CH, Harten J van, Wilson JHP, Danhof M, Brummelen P van, Breimer DD (1986) Nifedipine: Kinetics and hemodynamic effects in patients with liver cirrhosis after intravenous and oral administration. Clin Pharmacol Ther 40: 21–28
Gengo FM, Fagan SC, Krol G, Bernhard H (1987) Nimodipine disposition and haemodynamic effects in patients with cirrhosis and age-matched controls. Br J Clin Pharmacol 23: 47–53
Regard CG, Collste P, Edgar B et al. (1986) The effect of reduced hepatic function on the pharmacokinetics and hemodynamics of felodipine. Acta Pharmacol Toxicol 5 [Suppl V]: Abstract 900
Lasseter KC, Shamblen EC, Murdoch AA, Burkholder DE, Krol GJ, Taylor RJ, Vanov SK (1984) Steady-state pharmacokinetics of nitrendipine in hepatic insufficiency. J Cardiovasc Pharmacol 6: S977-S981
Williams RL, Mamelok RD (1980) Hepatic disease and drug pharmacokinetics. Clin Pharmacokinet 5: 528–547
MacMathúna P, O'Reilly T, Duenas-Laita A, Keeling PWN, Feely J (1986) The effect of calcium channel blockade with nifedipine on portal haemodynamics. Br J Clin Pharmacol 21: 578P-579P
Koshy A, Hadengue A, Lee SS, Jiron MI, Lebrec D (1987) Possible deleterious effect of nifedipine on portal hypertension in patients with cirrhosis. Clin Pharmacol Ther 42: 295–298
van Harten J, Lodewijks MThM, Guyt-Scholten JW, van Brummelen P, Breimer DD (1987) Gas chromatographic determination of nisoldipine and one of its metabolites in plasma. J Chromatogr Biomed Appl 423: 327–333
Gibaldi M, Perrier D (1982) Pharmacokinetics, 2nd edn. Marcel Dekker, New York
Goodnight JH (1982) in SAS user's guide: Statistics. SAS Institute Inc., Raleigh, NC
Raemsch K-D, Sommer J (1983) Pharmacokinetics and metabolism of nifedipine. Hypertension 5 [Suppl II]: 18–24
Baarnhielm C, Hansson G (1986) Oxidation of 1,4 dihydropyridines by prostaglandin synthetase and the peroxidic function of cytochrome P-450. Demonstration of a free radical intermediate. Biochem Pharmacol 35: 1419–1425
Wilkinson GR, Shand DG (1975) A physiological approach to hepatic drug clearance. Clin Pharmacol Ther 18: 377–389
Branch RA, James JA, Read AE (1977) The clearance of antipyrine and indocyanine green in normal subjects and in patients with chronic liver disease. Clin Pharmacol Ther 20: 81–89
Kleinbloesem CH, Brummelen P van, Danhof M, Faber H, Urquhart J, Breimer DD (1987) Rate of increase in the plasma concentration of nifedipine as a major determinant of its hemodynamic effects in humans. Clin Pharmacol Ther 41: 26–30
Kleinbloesem CH, Brummelen P van, Harten J van, Danhof M, Breimer DD (1985) Nifedipine: Influence of renal function on pharmacokinetic/hemodynamic relationship. Clin Pharmacol Ther 37: 563–574
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
van Harten, J., van Brummelen, P., Wilson, J.H.P. et al. Nisoldipine: Kinetics and effects on blood pressure and heart rate in patients with liver cirrhosis after intravenous and oral administration. Eur J Clin Pharmacol 34, 387–394 (1988). https://doi.org/10.1007/BF00542441
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00542441