Summary
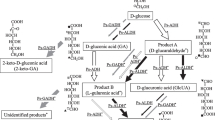
Mutants of Candida maltosa were isolated that lacked saccharopine reductase (lys9) and saccharopine dehydrogenase (lys1) and were able to accumulate α-aminoadipate-δ-semialdehyde (AASA) in the cell and excrete it into the culture medium. The effects of incubation time, lysine concentration, and carbon and nitrogen sources on AASA production were examined. In the presence of 15 g glucose/1, 1.25 g NH4H2PO4/l and 50 mg l-lysine/l in a minimal salt medium C. maltosa G285 (lys1) produced about 80–90 mg AASA/l during 48 h of growth. A simple and rapid procedure to isolate AASA from the medium using Dowex 50X4 is described.
Similar content being viewed by others
References
Aspen AJ, Meister A (1962) The preparation and some properties of α-aminoadipic-δ-semialdehyde (Δ1 acid). Biochemistry 1:600–605
Baginsky ML, Rodwell VW (1967) Metabolism of pipecolic acid in Pseudomonas species. V. Pipecolate oxidase and dehydrogenase. J Bacteriol 94:1034–1039
Basso LV, Rao DR, Rodwell VW (1962) Metabolism of pipecolic acid in a Pseudomonas species. II. Δ1 acid and α-aminoadipate acid-δ-semialdehyde. J Biol Chem 237:2239–2245
Bhattacharjee JK (1985) α-Aminoadipate pathway for the biosynthesis of lysine in lower eukaryotes. CRC Crit Rev Microbiol 12:131–151
Bhattacharjee JK, Sinha AK (1972) Relationship among the genes, enzymes and intermediates of the biosynthetic pathway of lysine in Saccharomyces. Mol Gen Genet 115:26–30
Böttcher F, Samsonova IA (1978) Rhodosporidium BANNO: Dosiseffektbeziehungen, Mutageneffektivität und Mutantespektrum bei der Induktion Auxotrophie-verursachender Mutationen durch ultraviolettes Licht und N-Methyl-N′-nitro-N-nitrosoaguanidin. Z Allg Mikrobiol 18:637–646
Kinzel JJ, Bhattacharjee JK (1982) Lysine biosynthesis in Rhodotorula glutinis: properties of pipecolic acid oxidase. J Bacteriol 151:1073–1077
Kinzel JJ, Winston MK, Bhattacharjee JK (1983) Role of l-lysine-α-ketoglutarate aminotransferase in catabolism of lysine as a nitrogen source for Rhodotorula glutinis. J Bacteriol 155:417–419
Larson RL, Sandine WD, Broquist HP (1963) Enzymic reduction of aminoadipic acid: relation to lysine biosynthesis. J Biol Chem 238:275–282
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–275
Schmidt H, Bode R, Lindner M, Birnbaum D (1985) Lysine biosynthesis in the yeast Candida maltosa: properties of some enzymes and regulation of the biosynthetic pathway. J Basic Microbiol 25:675–681
Schmidt H, Bode R, Birnbaum D (1988) A novel enzyme, l-lysine pyruvate aminotransferase, catalyzes the first step of lysine catabolism in Pichia guilliermondii. FEMS Microbiol Lett 49:203–206
Soda K, Misono H (1971) l-Lysine-α-ketoglutarate aminotransferase (Achromobacter liquidum). In: Tabor H, Tabor CW (eds) Methods in enzymology 17B. Academic Press, New York, pp 222–228
Soda K, Misono H, Yamamoto T (1968) l-Lysine-α-ketoglutarate aminotransferase. I. Identification of a product, Δ1 acid. Biochemistry 7:4102–4109
Tanaka A, Ohishi N, Fukui S (1967) Studies on the formation of vitamins and their function in hydrocarbon fermentation. Production of vitamin B6 by Candida albicans in hydrocarbon medium. J Ferment Technol 45:617–623
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Schmidt, H., Bode, R., Samsonova, I.A. et al. Production of α-aminoadipate-δ-semialdehyde by a mutant from Candida maltosa . Appl Microbiol Biotechnol 31, 463–466 (1989). https://doi.org/10.1007/BF00270777
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00270777