Summary
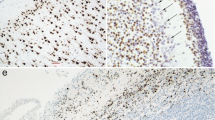
The histogenesis of the cerebral neocortex and early allocortex of the sheep has been described and, using an immunohistochemical technique, five plasma proteins have been identified in the telencephalic wall and their distribution followed during its differentiation. The development of the neocortex was studied from 18 days gestation, when the neural tube was still open, to 120 days, when the adult structure was established. A primordial plexiform layer was formed above the ventricular zone by 25 days and by 35 days this layer was divided by the differentiating cortical plate into an outer marginal zone and an inner subplate zone. The appearance of the subventricular and intermediate zones by 50 days gestation completed the formation of the neocortical layers. The differentiation of the allocortex was generally less advanced than the neocortex up to 40 days gestation, when the primordium of the pyramidal layer was beginning to develop.
The five plasma proteins identified, fetuin, α-fetoprotein, albumin, transferrin and α1-antitrypsin, are quantitatively the most important in the csf and plasma of the sheep fetus. Fetuin was the earliest plasma protein to be detected in the brain and it was also the most widespread; positive staining for this protein was seen in cells and fibres of all layers as they differentiated and could still be identified in some mature neurons at 120 days. α-Fetoprotein and albumin had a limited distribution, appearing in cells in the developing cortical plate for a short period early in gestation (35–40 days), but mainly confined to the ventricular zones later and barely detectable by 80 days gestation. Transferrin appeared to have a different distribution, being detected in fibres first in the primordial plexiform layer and then in the marginal and subplate zones, only later being identified in cells of the cortical plate. From their distribution it is suggested that fetuin and transferrin may play an important role in the differentiation of the cortex and the establishment of correct connections between fiber systems and migrating cells at certain stages of development. α1-Antitrypsin was only found in a few cells during a restricted period of gestation. All five plasma proteins were identified in precipitated csf and plasma at most ages examined, although at 18 days gestation albumin, transferrin and α1-antitrypsin and at 120 days, α-fetoprotein, could not be detected. The pattern of distribution of plasma proteins in the telencephalic wall suggests that they could originate either by uptake from csf and subsequent migration of protein containing cells or by local synthesis within some cells during a limited period of differentiation.
Similar content being viewed by others
References
Adamson ED (1982) The location and synthesis of transferrin in mouse embryos and teratocarcinoma cells. Dev Biol 91:227–234
All M, Mujoo K, Sahib MK (1983) Synthesis and secretion of alpha-fetoprotein and albumin by newborn rat brain cells in culture. Dev Brain Res 6:47–55
Aliau S, Marti J, Moretti J (1980) Levels of feto-proteins in fetal serum and extraembryonic fluids during ontogenesis in the ovine species. Int J Biochem: 49–54
Angevine JB Jr, Sidman RI (1961) Autoradiographic study of cell migration during histogenesis of cerebral cortex in the mouse. Nature 192:766–768
Åström KE (1967) On the early development of the isocortex in the fetal sheep. Prog Brain Res 26:1–59
Benno RH, Williams TH (1978) Evidence for intracellular localization of alpha-fetoprotein in the developing rat brain. Brain Res 142:182–186
Boulder Committec (1970) Embryonic vertebrate central nervous system: revised terminology. Anat Rec 166:257–261
Clausen PP (1980) Immunohistochemical demonstration of α1-antitrypsin in liver tissue. Acta Path Microbiol Scand Sect A 88:299–306
Cavanagh ME, Cornelis MEP, Dziegielewska KM, Luft AJ, Lai PCW, Lorscheider FF, Saunders NR (1982) Proteins in cerebrospinal fluid and plasma of fetal pigs during development. Dev Neurosci 5:492–502
Cavanagh ME, Cornelis MEP, Dziegielewska KM, Evans CAN, Møllgård K, Reynolds ML, Saunders NR (1983) Comparison of csf proteins in lateral ventricles and cisterna magna during early brain development of fetal sheep. Dev Brain Res 11:159–167
Cavanagh ME, Møllgård K (1984) An immunocytochemical study of the distribution of some plasma proteins within the developing forebrain of the pig with special reference to the neocortex. Dev Brain Res (in press)
Cavanagh ME, Warren A (1984) Uptake of plasma proteins from csf into the ventricular zone cells of the newborn rat. Accepted for publication J Physiol, 353:49 p
Dziegielewska KM (1982) Proteins in fetal csf and plasma. Ph D Thesis, University of London
Dziegielewska KM, Bock E, Cornelis MEP, Møllgård K, New H, Saunders NR (1983) Identification of fetuin in human and rat fetuses and in other species. Comp Biochem Physiol 76A: (no 2) 241–245
Dziegielewska KM, Evans CAN, Fossan A, Lorscheider FL, Malinowska DH, Møllgård K, Reynolds ML, Saunders NR, Wilkinson S (1980) Proteins in csf and plasma of fetal sheep during development. J Physiol 300:441–455
Dziegielewska KM, Evans CAN, Lorscheider FL, Malinowska DH, Møllgård K, Reynolds ML, Saunders NR (1981) Plasma proteins in fetal sheep brain: blood brain barrier and intracerebral distribution. J Physiol 318:239–250
Dziegielewska KM, Evans CAN, New H, Reynolds ML, Saunders NR (1984a) Plasma protein synthesis by fetal rat brain and choroid plexus. Int J Dev Neurosci 2 No 3:215–222
Dziegielewska KM, Reynolds ML, Sarantis MEP, Saunders NR (1984b) Plasma proteins in fetal sheep brain: correlation of immunohitochemical distribution and local synthesis. Accepted for publication J Physiol, 353:48 p
Dziegielewska KM, Saunders NR (1981) High concentration of α1-antitrypsin in fetal sheep csf and plasma. Comp Biochem Physiol 68B:307–311
Dziegielewska KM, Saunders NR (1982) Transferrin in fetal sheep cerebrospinal fluid and plasma during gestation. Comp Biochem Physiol 73A, 2:327–329
Evans CAN, Reynolds JM, Reynolds ML, Saunders NR, Segal MB (1974) The development of a blood brain barrier mechanism in fetal sheep. J Physiol 238:371–386
Jacobsen GK, Jacobsen M, Henriksen OB (1981) An immunohistochemical study of a series of plasma proteins in the early human conceptus. Oncodev Biol Med 2:399–410
Kourilsky FM, Burtin P (1968) Immunochemical difference between iron saturated and unsaturated human transferrin. Nature (London) 218:375–377
Lai PCW (1978) Alpha-fetoprotein in the rat and sheep. Ph D Thesis, University of Calgary
Mackiewicz A, Hejduk W, Breborowicz J (1978) The in vitro production of alpha-fetoprotein by human embryonic brain. Scand J Immunol (Suppl 8) 8:231–233
Marin-Padilla M (1978) Dual origin of the mammalian neocortex and evolution of the cortical plate. Anat Embryol 152:109–126
Marin-Padilla M (1983) Structural organization of the human cerebral cortex prior to appearance of the cortical plate. Anat Embryol 168:21–40
McKenna P (1984) Proteins in the amniotic and allantoic fluids of fetal pigs. Comp Biochem Physiol (in press)
Møllgård K, Jacobsen M (1984) Immunohistochemical identication of some plasma proteins in human embryonic and fetal forebrain with particular reference to the development of the neocortex. Dev Brain Res 13:49–63
Møllgård K, Jacobsen M, Jacobsen GK, Clausen PP, Saunders NR (1979) Immunohistochemical evidence for an intracellular localization of plasma proteins in human fetal choroid plexus and brain. Neurosci Lett 14:85–90
Møllgård K, Saunders NR (1975) Complex of tight junctions of epithelial and endothelial cells in the early fetal brain. J Neurocytol 4:453–468
Molliver ME (1982) Role of monoamines in the development of the neocortex. Neurosci Res Prog Bull 20:492–507
Molliver ME, Kostovic I, Van der Loos H (1973) The development of synapses in cerebral cortex of the human fetus. Brain Res 50:403–407
Molliver ME, Van der Loos H (1970) The ontogenesis of cortical circuitry: The spatial distribution of synapses in somaesthetic cortex of newborn dog. Ergbn Anat Entwicklungsgesch 42:1–54
Nowakowski RS, Rakic P (1981) The site of origin and route and rate of migration of neurons to the hippocampal region of the rhesus monkey. J Comp Neurol 196:129–155
Puck TT, Waldren A, Jones C (1968) Mammalian cell growth proteins. I. Growth stimulation by fetuin. Proc Natl Acad Sci USA 59:192–199
Raedler E, Raedler A, Feldhaus S (1980) Dynamical aspects of neocortical histogenesis in the rat. Anat Embryol 158:253–269
Rakic P, Nowakowski RS (1981) The time of origin of neurons in the hippocampal region of the rhesus monkey. J Comp Neurol 196:99–128
Reynolds ML, Evans CAN, Reynolds EOR, Saunders NR, Durbin GM, Wigglesworth JS (1979) Intracranial haemorrhage in the preterm sheep fetus. Early Human Dev 3/2:163–186
Reynolds ML, Møllgård K, Saunders NR (1983) The distribution of plasma proteins during early embryonic development in the sheep. Anat Embryol 168:227–240
Schultze HE, Heremans JF (1966) Molecular biology of human proteins with special reference to plasma proteins. In: Nature and Metabolism of Extracellular Proteins, Vol 1. Elsevier, Amsterdam
Sidman RL, Rakic P (1973) Neuronal migration with special reference to developing human brain. A review. Brain Res 62:1–35
Slade B, Budd S (1972) Localization of α-fetoprotein in fetal and newborn rabbits. Biol Neonate 21:309–320
Sternberger LA (1979) Immunocytochemistry, 2nd Ed. John Wiley and Sons, New York, pp 89–92
Toran-Allerand CD (1980) Coexistence of alpha-fetoprotein, albumin and transferrin immunoreactivity in neurons of the developing mouse brain. Nature (London) 288:733–735
Trojan J, Uriel J (1982) Immunocytochemical localization of alphafetoprotein (AFP) and serum albumin (ALB) in ecto, meso and endodermal tissue derivatives of the developing rat. Oncodev Biol Med 3:13–22
Uriel J, Trojan J, Dubouch P, Pineiro A (1982) Intracellular alphafetoprotein and albumin in the developing nervous system of the baboon. Patho. Biol 30:79–83
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Reynolds, M.L., Møllgård, K. The distribution of plasma proteins in the neocortex and early allocortex of the developing sheep brain. Anat Embryol 171, 41–60 (1985). https://doi.org/10.1007/BF00319053
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00319053