Summary
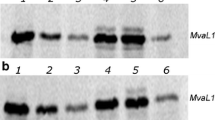
Two 30s ribosomal protein components, 30-4K and 30-7K, from E. coli K12 strain were clearly distinguished on the CMC column chromatogram from the corresponding protein components, 30-4B and 30-7B, from B strain. The 30-7K component was shown to correspond to the K-character.
A mutant strain of K12, W3637, had an altered 30s ribosomal protein component, 30-9W3637.
The characters of 30-4K, 30-7K, 30-9W3637 and str r were found to be cotransduced from W3637 to B strain by Plke phage in 16 out of 20 str r transductants. The 30-9W3637 and 30-4K components were separated from str r in 4 str r transductants. These results indicate that (1) neither 30-4 nor 30-9 is the protein whose mutational change leads to str r, and (2) the genes specifying the 30s ribosomal proteins, 30-4, 30-7, 30-9 and str are linked on the chromosome.
Similar content being viewed by others
Abbreviations
- CMC:
-
carboxymethyl cellulose
- str :
-
streptomycin
References
Apirion, D., and D. Schlessinger: Coresistance to neomycin and kanamycin by mutations in an Escherichia coli locus that affects ribosomes. J. Bact. 96, 768–776 (1968)
Cox, E. C., J. R. White, and J. G. Flaks: Streptomycin action and the ribosome. Proc. nat. Acad. Sci. (Wash.) 51, 703–709 (1964).
Davies, J. E.: Studies on the ribosomes of streptomycin-sensitive and resistant strains of Escherichia coli. Proc. nat. Acad. Sci. (Wash.) 51, 659–664 (1964).
— P. Anderson, and B. D. Davis: Inhibition of protein synthesis by spectinomycin. Science 149, 1096–1098 (1965).
Flaks, J. G., P. S. Leboy, E. A. Birge, and C. G. Kurland: Mutations and genetics concerned with the ribosome. Cold Spr. Harb. Symp. quant. Biol. 28, 623–631 (1966).
Hashimoto, K.: Streptomycin resistance in Escherichia coli analyzed by transduction. Genetics 45, 49–62 (1960).
Leboy, P. S., E. C. Cox, and J. G. Flaks: The chromosomal site specifying a ribosomal protein in Escherichia coli. Proc. nat. Acad. Sci. (Wash.) 52, 1367–1374 (1964).
Mayuga, C., D. Meier, and T. Wang: Escherichia coli: the K12 protein and the streptomycin region of the chromosome. Biochem. biophys. Res. Commun. 33, 203–206 (1968).
Osawa, S., E. Otaka, T. Itoh, and T. Fukui: Biosynthesis of 50s ribosomal subunit in Escherichia coli. J. molec. Biol. 46, 321–351 (1969).
Otaka, E., T. Itoh, and S. Osawa: Ribosomal proteins of bacterial cells: strain and species-specificity. J. molec. Biol. 33, 93–107 (1968).
Ozaki, M., S. Mizushima, and M. Nomura: Identification and functional characterization of the protein controlled by streptomycin-resistant locus in E. coli. Nature (Lond.) 222, 333–339 (1969).
Rosset, R., and L. Gorini: A ribosomal ambiguity mutation. J. molec. Biol. 39, 95–112 (1969).
tanaka, Y., and H. Kaji: The role of ribosomal protein for the binding of dihydrostreptomycin to ribosomes. Biochem. biophys. Res. Commun. 32, 313–319 (1968).
Traub, P., and M. Nomura: Streptomycin resistance mutation in Escherichia coli: altered ribosomal protein. Science 160, 198–199 (1968).
Waller, J. P., and J. I. Harris: Studies on the composition of the protein from Escherichia coli ribosomes. Proc. nat. Acad. Sci. (Wash.) 47, 18–23 (1961).
Watanabe, I.: The effect of ultraviolet light on the production of bacterial virus protein. J. gen. Physiol. 40, 521–531 (1957).
Author information
Authors and Affiliations
Additional information
Communicated by H. G. Wittmann
Rights and permissions
About this article
Cite this article
Takata, R., Dekio, S., Otaka, E. et al. Genetic studies of the ribosomal proteins in Escherichia coli . Molec. Gen. Genet. 105, 113–121 (1969). https://doi.org/10.1007/BF00445680
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00445680