Summary
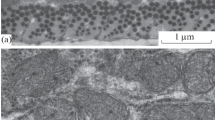
Scanning electron microscopy was used to investigate the morphological changes of the tail musculature of the metamorphosing anuran tadpole, attention being focused on phagocytosis by macrophages. Muscle fibers were stained en bloc with silver and freeze-fractured during dehydration, or torn after drying. Samples were sputter-coated with gold-palladium and observed in both secondary electron- and back-scattered electron modes with a scanning electron microscope.
Various cells were identified by the methods of secondary electron- and back-scattered electron images. Some macrophages lying between muscle fibers at prometamorphic stages possessed numerous finger-like projections and well-developed ruffles. During degeneration of muscle fibers macrophages collected in the degenerating region and invaded the space between the disordering myofibrils. In advanced stages the numbers of macrophages clearly increased on or around the degenerating muscle fibers. At the climactic stage fragmented muscles were entrapped and then engulfed by the macrophages. With the completion of phagocytosis, the macrophages became globular with reduction of the ridge-like ruffles. Macrophages may play a role not only in scavenging the fragmented muscle fibers, but also using their long processes in active formation of the fragments.
Similar content being viewed by others
References
Abercrombie M, Heaysman JEM, Pegrum SM (1970) The locomotion of fibroblasts in culture. I. Movements of the leading edge. Exp Cell Res 59:393–398
Bard JBL, Hay ED, Meller SM (1975) Formation of the endothelium of the avian cornea: A study of cell movement in vivo. Dev Biol 42:334–361
Becker RP, Sogard M (1979) Visualization of subsurface structures in cells and tissues by backscattered electron imaging. In: Johari O, Becker RP (eds) Scanning electron microscopy. Vol 2, SEM Inc, Chicago, pp 835–870
Bielschowsky M (1919) Einige Bemerkungen zur normalen und pathologischen Histologie des Schweifund Linsenkerns. J Psychol Neurol 25:1–11
Boug TK, Caulfield JB (1980) Morphology of connective tissue in skeletal muscle. Tissue Cell 12:197–207
Boyde A, Williams JCP (1968) Surface morphology of frog striated muscle as prepared for and examined in the scanning electron microscope. J Physiol 197, pp 10–11
Carr KE, Toner PG (1979) Scanning electron microscopy of macrophages: A bibliography. In: Johari O, Becker RP (eds) Scanning electron microscopy. Vol 3, SEM Inc, Chicago, pp 637–644
De Nee PB, Frederickson RG, Pope RS (1977) Heavy metal staining of paraffin, epoxy and glycol methacrylate embedded biological tissue for scanning electron microscope histology. In: Johari O, Becker RP (eds) Scanning electron microscopy. Vol 2, IIT Res Inst, Chicago, pp 83–92
Desaki J, Uehara Y (1981) The overall morphology of neuro-muscular junctions as revealed by scanning electron microscopy. J Neurocytol 10:101–110
Dichiara JF, Rowley PP, Ogilvie RW (1980) Backscatter electron imaging (BEI) of paraffin sections stained with heavy metal histopathologic stains, with observations on some variable encountered in BEI. In: Johari O, Becker RP (eds) Scanning electron microscopy. Vol 3, SEM Inc, Chicago, pp 181–188
Fox H (1975) Aspects of tail muscle ultrastructure and its degeneration in Rana temporaria. J Embryol Exp Morphol 34:191–207
Fujita T, Kashimura M (1981) The “reticulo-endothelial system” reviewed by scanning electron microscopy. Biomed Res 2 Suppl:159–171
Gnepp DR, Green FHY (1979) Scanning electron microscopy of collecting lymphatic vessels and their comparison to arteries and veins. In: Johari O, Becker RP (eds) Scanning electron microscopy. Vol 3, SEM Inc, Chicago, pp 757–762
Greer MH, Greer RT (1969) Progressive surface morphological changes of muscle fibers. In: Johari O (ed) Scanning electron microscopy. IIT Res Inst, Chicago, p 177–183
Horguchi T, Watanabe K (1984) Morphometric study on vascularization in the tail musculature of the anuran tadpole by scanning electron microscopy. I. Prometamorphic stage. Anat Rec 208:329–335
Horiguchi T, Sasaki F, Takahama H, Watanabe K (1984) Identification of cells by backscattered electron imaging of silver stained bulk tissue in scanning electron microscopy. Stain Technol 59:143–148
Karnovsky MJ (1965) A formaldehyde-glutaraldehyde fixative of high osmolality for use in electron microscopy. J Cell Biol 27:137a-138a
Kerr JFR, Harmon B, Searle J (1974) An electron-microscope study of cell deletion in the anuran tadpole tail during spontaneous metamorphosis with special reference to apoptosis of striated muscle fibers. J Cell Sci 14:571–585
Lim DJ (1971) Scanning electron microscopic observation on nonmechanically cryofractured biological tissue. In: Johari O, Corvin I (eds) Scanning electron microscopy. IIT Res Inst, Chicago, pp 257–264
Mayer S (1886) Die sogenannten Sarkoplasten. Anat Anz 1:231–235
McCallister LP, Hadek R (1970) Transmission electron microscopy and stereo ultrastructure of the T system in frog skeletal muscle. J Ultrastruct Res 33:360–368
Muto M, Fujita T (1977) Phagocytotic activites of the Kupffer Cell: A scanning electron microscope study. In: Wisse E, Knook DL (eds) Kupffer cells and other liver sinusoidal cells. Elsevier/North-Holland, Biomedical Press, Amsterdam, pp 109–119
Nelson GA, Revel JP (1975) Scanning electron microscopic study of cell movements in the corneal endothelium of the avian embryo. Dev Biol 42:315–333
Ogura K, Laudate A (1980) Comparative observation with a light microscope and an SEM in backscattered electron mode. In: Johari O (ed) Scanning electron microscopy. Vol 1, SEM Inc, Chicago, pp 233–238
Polliack A, Gordon S (1975) Scanning electron microscopy of murine macrophages. Lab Invest 33: Lab Invest 33:469–477
Revel JP (1974) Scanning electron microscope studies of cell surface morphology and labeling, in situ and in vitro. In: Johari O, Corvin I (eds) Scanning electron microscopy. IIT Res Inst Chicago, pp 541–548
Rheuben MP, Reese TS (1978) Three-dimensional structure and membrane specializations of moth excitatory neuromuscular synapse. J Ultrastruct Res 65:95–111
Sawada H, Ishikawa H, Yamada E (1978) High resolution scanning electron microscopy of frog sartorius muscle. Tissue Cell 10:179–190
Shimada Y, Fischman DA (1975) Scanning electron microscopy of nerve-muscle contacts in embryonic cell culture. Dev Biol 43:42–61
Stickland NC (1982) Scanning electron microscopy of prenatal muscle development in the mouse. Anat Embryol 164:379–385
Taylor AC, Kollros JJ (1946) Stages in the normal development of Rana pipiens larvae. Anat Rec 94:7–23
Tokunaga M, Tokunaga J, Niimi M (1981) Leukocyte and macrophage movements under phagocytosis. Biomed Res 2 Suppl:13–22
Trinkaus JP, Betchaku T, Krulikowski LS (1971) Local inhibition of ruffling during contact inhibition of cell movement. Exp Cell Res 64:291–300
Varute AT (1971) Histoenzymorphology of β-glucuronidase in the resorbing tails of tadpoles of Rana tigrina at metamorphosis. Acta Histochem 41:306–324
Vriend RA, Geissinger HD (1980) An improved direct inter-microscopic (LM → SEM → TEM) correlative procedure for the examination of mammalian skeletal muscle. J Microsc 120:53–64
Watanabe K, Sasaki F (1974) Ultrastructural changes in the tail muscles of anuran tadpoles during metamorphosis. Cell Tissue Res 155:321–336
Weber R (1964) Ultrastructural changes in regressing tail muscles of Xenopus larvae at metamorphosis. J Cell Biol 22:481–487
Weiss P (1961) Guiding principles in cell locomotion and cell aggregation. Exp Cell Res Suppl 8:260–281
Wroblewski R, Roomans GM, Jansson E, Edström L (1978) Electron probe X-ray microanalysis of human muscle biopsies. Histochemistry 55:281–292
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Watanabe, K., Horiguchi, T. & Sasaki, F. Scanning electron microscopy of macrophages in the tail musculature of the metamorphosing anuran tadpole, Rana japonica . Cell Tissue Res. 241, 545–550 (1985). https://doi.org/10.1007/BF00214574
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00214574