Summary
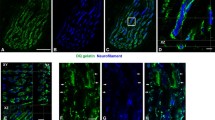
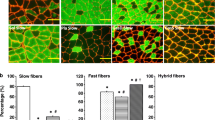
The fine structure of the M-band in soleus (SOL) and extensor digitorum longus (EDL) muscles in newborn and four-week-old rats was studied using electron-microscopic techniques. In newborn rats, all myotubes and fibres in both muscles had an identical myofibrillar appearance. A five-line M-band pattern was seen in longitudinal sections and distinct M-bridges in cross-sections. The Z-discs were of medium width. On the other hand, in four-week-old rats, different muscle fibre types were observed on the basis of their myofibrillar pattern. In SOL two fibre types were distinguished in longitudinal sections. One had a four-line M-band pattern and very broad Z-discs, whereas the other type had five lines in the M-band and broad Z-discs. In EDL, three different myofibrillar patterns were observed. The M-bands were composed of three, four or five lines. Fibres had either thin, broad or medium Z-disc widths, respectively. In cross-sections of the SOL muscle one group of fibres showed indistinct M-bridges, whereas distinct M-bridges were seen in the other fibres and in all observed EDL muscle fibres. We conclude that initially there seems to be a single intrinsic program for M-band genesis; this program becomes modified upon the induction of functionally differentiated fibres.
Similar content being viewed by others
References
Anversa P, Olivetti G, Bracchi P-G, Loud AV (1981) Postnatal development of the M-band in rat cardiac myofibrils. Circ Res 48:561–568
Bennett MR (1983) Development of neuromuscular synapses. Physiol Rev 63:915–1048
Brooke MH, Williamson E, Kaiser KK (1971) The behavior of four fiber types in developing and reinnervated muscle. Arch Neurol 25:360–366
Brown MC, Booth CM (1983) Postnatal development of the adult pattern of motor axon distribution in rat muscle. Nature 304:741–742
Carlsson E, Kjörell U, Thornell L-E, Strehler EE, Lambertsson A (1982) Differentiation of the myofibrils and the intermediate filament system during postnatal development of the rat hearts. Eur J Cell Biol 27:62–73
Close RI (1972) Dynamic properties of mammalian skeletal muscle. Physiol Rev 52:129–197
De Coster W, De Reuck J, Vander Eecken H (1984) The use of semiautomatic morphometry in the study of normal rat gastrocnemius muscle fibers. Acta Neuropathol (Berl) 64:108–113
Eisenberg BR (1983) Quantitative ultrastructure of mammalian skeletal muscle. In: Peachey LD, Adrian RH, Geiger SR (eds) Handbook of Physiology Skeletal Muscle. Am Phys Soc, Bethesda, Maryland, pp 73–112
Eisenberg BR, Salmons S (1981) The reorganization of subcellular structure in muscle undergoing fast-to-slow type transformation. Cell Tissue Res 220:449–471
Eisenberg BR, Brown JMC, Salmons S (1984) Restoration of fast muscle characteristics following cessation of chronic stimulation. The ultrastructure of slow-to-fast transformation. Cell Tissue Res 238:221–230
Fairfield J (1948) Effects of cold on infant rats: Body temperature, oxygen consumption, electrocardiograms. Am J Physiol 128:514
Fischman DA (1967) An electron microscope study of myofibril formation in embryonic chick skeletal muscle. J Cell Biol 32:557–575
Gauthier GF (1974) Some ultrastructural and cytochemical features of fiber populations in the soleus muscle. Anat Rec 180:551–564
Gauthier GF (1979) Ultrastructural identification of muscle fiber types by immunocytochemistry. J Cell Biol 82:391–400
Grove BK, Kurer V, Lehner C, Doetschman TC, Perriard J-C, Eppenberger HM (1984) A new 185,000-dalton skeletal muscle protein detected by monoclonal antibodies. J Cell Biol 98:518–524
Hanzlikova V, Schiaffino S (1973) Studies on the effect of denervation in developing muscle. III. Diversification of myofibrillar structure and origin of the heterogeneity of muscle fiber types. Z Zellforsch 147:75–85
Kelly AM (1983) Emergence of specialization in skeletal muscle. In: Peachey LD, Adrian RH, Geiger SR (eds) Handbook of Physiology Skeletal Muscle. Am Phys Soc, Bethesda, Maryland, pp 507–537
Knappeis GG, Carlsen F (1968) The ultrastructure of the M line in skeletal muscle. J Cell Biol 38:202–211
Kugelberg E (1976) Adaptive transformation of rat soleus motor units during growth. Histochemistry and contraction speed. J Neurol Sci 27:269–289
Lazarides E (1980) Intermediate filaments as mechanical integrators of cellular space. Nature 283:249–256
Luther P, Squire J (1978) Three-dimensional structure of the vertebrate muscle M-region. J Mol Biol 125:313–324
Luther P, Munro PMG, Squire JM (1981) Three-dimensional structure of the vertebrate muscle A-band. III. M-region structure and myosin filament symmetry. J Mol Biol 151:703–730
Masaki T, Takaiti O (1974) M-protein. J Biochem (Tokyo) 75:367–380
Morimoto K, Harrington WF (1972) Isolation and physical chemical properties of an M-line protein from skeletal muscle. J Biol Chem 247:3052–3061
Page SG (1965) A comparison of the fine structure of frog slow and twitch fibers. J Cell Biol 26:477–497
Peng HB, Wolosewick JJ, Cheng P-C (1981) The development of myofibrils in cultured muscle cells: A whole-mount and thinsection electron microscopic study. Dev Biol 88:121–136
Pepe FA (1967) The myosin filament. I. Structural organization from antibody staining observed in electron microscopy. J Mol Biol 27:203–225
Pepe FA (1975) Structure of muscle filaments from immunohistochemical and ultrastructural studies. J Histochem Cytochem 23:543–562
Pepe FA (1983) Immunological techniques in fluorescence and electron microscopy applied to skeletal muscle fibers. In: Peachey LD, Adrian RH, Geiger SR (eds) Handbook of Physiology Skeletal Muscle. Am Phys Soc Bethesda, Maryland pp 113–141
Redfern PA (1970) Neuromuscular transmission in newborn rats. J Physiol 209:701–709
Riley DA (1977) Multiple innervation of fiber types in the soleus muscles of postnatal rats. Exp Neurol 56:400–409
Rubinstein NA, Kelly AM (1981) Development of muscle fiber specialization in the rat hindlimb. J Cell Biol 90:128–144
Salmons S, Gale DR, Sréter FA (1978) Ultrastructural aspects of the transformation of muscle fibre type by long term stimulation: changes in Z discs and mitochondria. J Anat 127:17–31
Schiaffino S (1972) Differentiation of the contractile apparatus in vertebrate skeletal muscles. Boll Zool 39:139–158
Shafiq SA, Asiedu SA, Milhorat AT (1972) Effect of neonatal neurectomy on differentiation of fiber types in rat skeletal muscle. Exp Neurol 35:529–540
Sjöström M, Squire JM (1977a) Fine structure of the A-band in cryosections. The structure of the A-band of human skeletal muscle fibres from ultra-thin cryo-sections negatively stained. J Mol Biol 109:49–68
Sjöström M, Squire JM (1977b) Cryo-ultramicrotomy and myofibrillar fine structure: A review. J Microsc 111:239–278
Sjöström M, Kidman S, Henriksson Larsén K, Ängquist K-A (1982a) Zand M-band appearance in different histochemically defined types of human skeletal muscle fibers. J Histochem Cytochem 30:1–11
Sjöström M, Ängquist K-A, Bylund A-C, Fridén J, Gustavsson L, Scherstén T (1982b) Morphometric analyses of human muscle fiber types. Muscle Nerve 5:538–553
Squire J (1981) The Structural Basis of Muscular Contraction. Plenum Press, New York London
Squire J, Edman A-C, Freundlich A, Harford J, Sjöström M (1982) Muscle structure, cryo-methods and image analysis. J Microsc 125:215–225
Strehler EE, Carlsson E, Eppenberger HM, Thornell L-E (1983) Ultrastructural localization of M-band proteins in chicken breast muscle as revealed by combined immunocytochemistry and ultramicrotomy. J Mol Biol 166:141–158
Taniguchi M, Ishikawa H (1982) In situ reconstitution of myosin filaments within the myosin-extracted myofibril in cultured skeletal muscle cells. J Cell Biol 92:324–332
Thornell L-E, Sjöström M (1975) The myofibrillar M-band in the cryo-section analysis of section thickness. J Microsc 104:263–269
Thornell L-E, Sjöström M, Ringqvist M (1976) Attempts to correlate histochemical and ultrastructural features of individual skeletal muscle fibers. J Ultrastruct Res 57:224
Thornell L-E, Carlsson E, Kugelberg E, Grove BK, Eppenberger HM (1985) New aspects on the structure and composition on the myofibrillar M-band. J Muscle Res Cell Motil 6:81
Traeger L, Goldstein MA (1983) Thin filaments are not of uniform length in rat skeletal muscle. J Cell Biol 96:100–103
Trinick J, Lowey S (1977) M-protein from chicken pectoralis muscle: isolation and characterization. J Mol Biol 113:343–368
Turner DC, Wallimann T, Eppenberger HM (1973) A protein that binds specifically to the M-line of skeletal muscle is identified as the muscle form of creatine kinase. Proc Natl Acad Sci USA 70:702–705
Wallimann T, Schlösser T, Eppenberger HM (1984) Function of M-line-bound CK as intramyofibrillar ATP regenerator at the receiving end of the CP shuttle. J Biol Chem 259:5238
Wang K, Ramirez-Mitchell R (1983) A network of transverse and longitudinal intermediate filaments is associated with sarcomeres of adult vertebrate skeletal muscle. J Cell Biol 96:562–570
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Carlsson, E., Thornell, L.E. Diversification of the myofibrillar M-band in rat skeletal muscle during postnatal development. Cell Tissue Res. 248, 169–180 (1987). https://doi.org/10.1007/BF01239978
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01239978