Summary
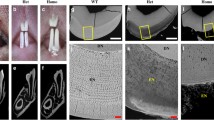
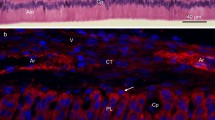
Experiments were designed to detect and determine the biosynthetic behavior of enamel proteins in Syrian Golden hamsters. Enamel matrix proteins were extracted from 3-day-old postnatal first molar tooth organs. Labeling pulse/chase experiments with [35S]-methionine followed by light microscopic autoradiography, or polyacrylamide slab gel electrophoresis and fluorography, showed the synthesis of epithelial-specific gene products. Synthesis and secretion of enamel proteins required approximately 30 min under thesein vitro organ culture conditions; both enamelin and amelogenin proteins were synthesized and secreted into the forming extracellular matrix. Amelogenin proteins were secreted initially and rapidly degraded into increasingly smaller polypeptides. In contrast, enamelin proteins were secreted at a slower rate and remained more or less stable over the duration of the experiment. The specific activities of both classes of proteins increased over a 6-hour synthesis period, indicating the accumulation of both proteins into the forming extracellular matrix. Comparisons of the kinetics of formation and posttranslational processing of enamelin and amelogenin are consistent with the presence of possibly two different gene products in hamster secretory ameloblasts.
Similar content being viewed by others
References
Slavkin H, Trump G, Schonfeld S, Brownell A, Sorgente N, Lee-Own V (1978) Epigenetic regulation of enamel protein synthesis during epithelial-mesenchymal interactions. In: Saxen L, Weiss L (eds) Cell interactions in differentiation, Academic Press, New York, pp 209–226
Frank RM (1979) Tooth enamel: Current state of the art. J Dent Res 58(B):684–693
Eastoe JE (1979) Enamel protein chemistry—past, present and future. J Dent Res 58(B):753–763
Guenther HC, Croissant RD, Schonfeld SE, Slavkin HC (1977) Identification of four extracellular-matrix enamel proteins during embryonic-rabbit tooth-organ development. Biochem J 163:591–603
Fukae M, Shimizu M (1974) Studies on the proteins of developing bovine enamel. Arch Oral Biol 19:381–386
Shimizu M, Tanabe T, Fukae M (1979) Proteolytic enzyme in porcine immature enamel. J Dent Res 58(B):782–789
Chrispens J, Weliky B, Bringas P, Slavkin H (1979) Proenamel-enamel-polypeptide: A concept. J Dent Res 58(B):988–990
Slavkin HC, Weliky B, Stellar W, Slavkin MD, Zeichner-Gancz M, Bringas P Jr, Hyatt-Fischer H, Shimizu M, Fukae M (1979) Ameloblast differentiation: Protein synthesis and secretion in fetal New Zealand white rabbit molar tooth organs and isolated epithelialin vitro. J Biol Buccale 6:309–326
Termine JD, Torchia DA, Conn KM (1979) Enamel matrix: Structural proteins. J Dent Res 58(B):773–778
Termine JD, Belcourt AB, Christner PJ, Conn KM, Nylen MU (1980) Properties of dissociatively extracted fetal tooth matrix proteins. I. Principle molecular species in developing bovine enamel. J Biol Chem 255:9760–9768
Saski S, Shimokawa H (1979) Enamel proteins: Biosynthesis and chemistry. J Dent Res 58(B):765–771
Lyaruu DM, Belcourt A, Fincham AG, Termine JD (1982) Neonatal hamster molar tooth development: Extraction and characterization of amelogenins, enamelins, and soluble dentin proteins. Calcif Tissue Int 34:86–96
Yamada M, Bringas P Jr, Grodin M, MacDougall M, and Slavkin HC (1980) Developmental comparisons of murine secretory amelogenesis in vivo, as xenografts on the chick chorio-allantoic membrane, and in vitro. Calcif Tissue Int 31:161–171
Slavkin HC, Flores P, Bringas P, Bavetta LA (1970) Epithelial-mesenchymal interactions during odontogenesis. I. Isolation of several intercellular matrix low molecular weight methylated RNAs. Dev Biol 23:276–296
Slavkin HC, Mino W, Bringas P Jr (1976) The biosynthesis and secretion of precursor enamel protein by ameloblasts as visualized by autoradiography after tryptophan administration. Anat Rec 185:289–312
Patzelt C, Labrecque AD, Duguid JR, Carroll RJ, Keim PS, Heinrikson RL, Steiner DF (1978) Detection and kinetic behavior of preproinsulin in pancreatic islets. Proc Natl Acad Sci USA 75:1260–1264
Bradford MM (1976) A rapid and sensitive method for the quantitative of microgram quantities of protein utilizing the principles of protein-dye binding, Anal Biochem 72:248–254
Davis BJ (1964) Disc Electrophoresis—II Method and application to human serum proteins. Ann NY Acad Sci 121:404–427
Ornstein L (1964) Disc electrophoresis—I Background and theory. Ann NY Acad Sci 121:321–348
Bonner WM, Laskey RA (1974) A film detection method for tritium-labeled proteins and nucleic acid in polyacrylamide gels. Eur J Biochem 46:83–88
Laskey RA, Mills AD (1975) Quantitative film detection of3H and14C in polyacrylamide gels by fluorography. Eur J Biochem 56:335–341
Fincham AG (1979) The amelogenin problem: A comparison of purified enamel matrix proteins. Calcif Tissue Int 27:65–73
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Zeichner-David, M., Slavkin, H.C., Lyaruu, D.M. et al. Biosynthesis and secretion of enamel proteins during hamster tooth development. Calcif Tissue Int 35, 366–371 (1983). https://doi.org/10.1007/BF02405060
Issue Date:
DOI: https://doi.org/10.1007/BF02405060