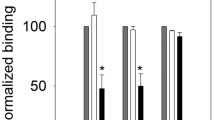
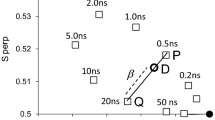
Summary
To elucidate further the structure and molecular dynamics of the epidermal growth factor receptor, temperature-dependent aggregation and extracellular protrusion of the epidermal growth factor receptor in isolated plasma membranes from A431 cells were examined by fluorescence energy-transfer techniques. Epidermal growth factor was labeled at the amino terminus with either fluorescein isothiocyanate or tetramethylrhodamine isothiocyanate. A radionuclide receptor displacement assay demonstrated the bioactivity of these derivatives. Aggregation of the epidermal growth factor receptor was measured by determining the increase in fluorescence energy transfer between receptorbound fluorescein and tetramethylrhodamine-labeled epidermal growth factor. Energy transfer between receptor-bound fluorescent derivatives was reversibly greater at 37 than 4°C, indicating temperature-dependent aggregation of the receptor. The extracellular protrusion of the epidermal growth factor receptor was calculated from the magnitude of energy transfer between receptorbound fluorescein labeled epidermal growth factor and 5-(N-dodecanoylamino)-eosin partitioned into the lipid membrane at 4 and 37°C. No significant change in the distance of closest approach between the N-terminus of epidermal growth factor and the plasma membrane was observed at 4°C (69±2 Å) and 37°C (67±2 Å). Thus, the extracellular protrusion of the occupied epidermal growth factor receptor did not change detectably upon receptor aggregation.
Similar content being viewed by others
References
Basu, M., Sen-Majumdar, A., Basu, A., Murthy, U., Das, M. 1986. Regulation of kinase and intermolecular bonding in intact and truncated epidermal growth factor receptor.J. Biol. Chem. 261:12879–12882
Berman, H.A., Yguerabide, J., Taylor, P. 1980. Fluorescence energy transfer on acetylcholinesterase: Spatial relationship between peripheral site and active center.Biochemistry 19:2226–2235
Biswas, R., Basu, M., Sen-Majumdar, A., Das, M. 1985. Intrapeptide autophosphorylation of the epidermal growth factor receptor: Regulation of kinase catalytic function by receptor dimerization.Biochemistry 24:3795–3802
Boni-Schnetzler, M., Pilch, P.F. 1987. Mechanism of epidermal growth factor receptor autophosphorylation and high-affinity binding.Proc. Natl. Acad. Sci. USA 84:7832–7836
Carpenter, G., Cohen, S. 1976.125I-labeled human epidermal growth factor binding, internalization, and degradation in human fibroblasts.J. Cell Biol. 71:159–171
Carpenter, G., Cohen, S. 1979. Epidermal growth factor.Annu. Rev. Biochem. 48:193–216
Carpenter, G., King, L., Jr., Cohen, S. 1979. Rapid enhancement of protein phosphorylation in A431 cell membrane preparations by epidermal growth factor.J. Biol. Chem. 254:4884–4891
Carpenter, G., Zendegui, J.G. 1986. Epidermal growth factor, its receptor, and related proteins.Exp. Cell Res. 164:1–10
Carraway, K.L., Koland, J.G., Cerione, R.A. 1989. Visualization of epidermal growth factor (EGF) receptor aggregation in plasma membranes by fluorescence resonance energy transfer.J. Biol. Chem. 264:8699–8707
Cassel, D., Glaser, L. 1982. Proteolytic cleving of epidermal growth factor receptor: A Ca2+-dependent, sulfhydryl-sensitive proteolytic system in A431 cells.J. Biol. Chem. 257:9845–9848
Chen, R.F. 1965. Fluorescence quantum yield measurements: Vitamin B6 compounds.Science 150:1593–1595
Chen, W.S., Lazar, C.S., Poenie, M., Tsien, R.Y., Gill, G.N., Rosenfeld, M.G. 1987. Requirement for intrinsic protein tyrosine kinase in the immediate and late actions of the EGF receptor.Nature (London) 328:820–823
Cochet, C., Kashles, O., Chanbaz, E.M., Borrello, I., King, C.R., Schlessinger, J. 1988. Demonstration of epidermal growth factor-induced receptor dimerization in living cells using a chemical covalent cross-linking agent.J. Biol. Chem. 263:3290–3295
Cohen, S., Ushiro, H., Stoscheck, C., Chinkers, M. 1982. A native 170,000 epidermal growth factor receptor-kinase complex from shed plasma membrane vesicles.J. Biol. Chem. 257:1523–1531
Dale, R.E., Eisinger, J., Blumberg, W.E. 1979. The orientation factor in intramolecular energy transfer.Biophys. J. 26:161–194
Fanger, B.O., Austin, K.S., Earp, H.S., Cidlowski, J.A. 1986. Cross-linking of epidermal growth factor receptors in intact cells: Detection of initial stages of receptor clustering and determination of molecular weight of high-affinity receptors.Biochemistry 25:6414–6420
Fung, B.K.-K., Stryer, L. 1978. Surface density determination in membranes by fluorescence energy transfer.Biochemistry 17:5241–5248
Hillman, G.M., Schlessinger, J. 1982. Lateral diffusion of epidermal growth factor complexed to its surface receptors does not account for the thermal sensitivity of patch formation and endocytosis.Biochemistry 21:1667–1672
Holowka, D., Baird, B. 1983a. Structural studies on the membrane-bound immunoglobulin E-receptor complex: I. Characterization of large plasma membrane vesicles from rat basophilic leukemia cells and insertion of amphipathic fluorescent probes.Biochemistry 22:3466–3474
Holowka, D., Baird, B. 1983b. Structural studies on the membrane-bound immunoglobulin E-receptor complex: II. Mapping of distances between sites on IgE and the membrane surface.Biochemistry 22:3475–3484
Honegger, A.M., Dull, T.J., Felder, S., Van Obberghen, E., Bellot, F., Szapary, D., Schmidt, A., Ullrich, A., Schlessinger J. 1987. Point mutation at the ATP binding site of EGF receptor abolishes protein-tyrosine kinase activity and alters cellular routing.Cell 51:199–209
Koland, J.G., Cerione, R.A. 1988. Growth factor control of epidermal growth factor receptor kinase activity via an intramolecular mechanism.J. Biol. Chem. 263:2230–2237
Laemmeli, U.K. 1970. Cleavage of structural proteins during the assembly of the head of bacteriophage T4.Nature (London) 227:680–685
Lowry, O.H., Rosebrough, N.J., Farr, A.L., Randall, R.J. 1951. Protein measurement with the Folin phenol reagent.J. Biol. Chem. 193:265–275
Moolenaar, W.H., Bierman, A.J., Tilly, B.C., Verlaan, I., Defize, L.H.K., Honegger, A.M., Ullrich, A., Schlessinger, J. 1988. A point mutation at the ATP-binding site of the EGF-receptor abolishes transduction.EMBO J 7:707–710
Parker, C.A., Rees, W.T. 1960. Correction of fluorescence spectra and measurement of fluorescence quantum efficiency.Analyst 85:587–600
Santon, J.B., Cronin, M.T., Macleod, C.L., Mendelsohn, J., Masui H., Gill, G.N. 1986. Effects of epidermal growth factor receptor concentration on tumorigenicity of A431 cells in nude mice.Cancer Res. 46:4701–4705
Savage, C.R., Cohen, S. 1972. Epidermal growth factor and a new derivative.J. Biol. Chem. 247:7609–7611
Shecter, Y., Schlessinger, J., Jacobs, S., Chang, K.J., Cuatrecasas, P. 1978. Fluorescent labeling of hormone receptors in viable cells: Preparation and properties of highly fluorescent derivatives of epidermal growth factor and insulin.Proc. Natl. Acad. Sci. USA 75:2135–2139
Schlessinger, J. 1986. Allosteric regulation of the epidermal growth factor receptor kinase.J. Cell. Biol. 103:2067–2072
Shaklai, N., Yguerabide, J., Ranney, H.M. 1977. Interaction of hemoglobin with red blood cell membranes as shown by a fluorescent chromophore.Biochemistry 16:5585–5592
Stryer, L., Thomas, D.D., Carlsen, W.F. 1982. Fluorescence energy transfer measurements of distances in rhodopsin and the purple membrane protein.Methods Enzymol. 81:668–678
Taylor, J.M., Mitchel, W.M., Cohen, S. 1972. Epidermal growth factor.J. Biol. Chem. 247:5928–5934
Ullrich, A., Coussens, L., Hayflick, J.S., Dull, T.J., Gray, A., Tam, A.W., Lee, J., Yarden, Y., Libermann, T.A., Schlessinger, J., Downward, J., Mayes, E.L.V., Whittle, N., Waterfield, M.D., Seeburg, P.H. 1984. Human epidermal growth factor receptor cDNA sequence and aberrant expression of the amplified gene in A431 epidermoid carcinoma cells.Nature (London) 309:418–425
Umberger, J.Q., LaMer, V.K. 1945. The kinetics of diffusion controlled molecular and ionic reactions in solution as determined by measurements of the quenching of fluorescence.J. Am. Chem. Soc. 67:1099–1109
Weber, K., Pringle, J.R., Osborn, M. 1972. Measurement of molecular weights by electrophoresis on SDS-acrylamide gel.Methods Enzymol. 26:3–27
Weber, W., Bertics, P.J., Gill, G.N. 1984. Immunoaffinity purification of the epidermal growth factor receptor: Stoichiometry of binding and kinetics of self-phosphorylation.J. Biol. Chem. 259:14631–14636
Wolber, P.K., Hudson, B.S. 1979. An analytic solution to the forster energy transfer problem in two dimensions.Biophys. J. 28:197–210
Yarden, Y., Schlessinger, J. 1987a. Self-phosphorylation of epidermal growth factor receptor: Evidence for a model of intermolecular allosteric activation.Biochemistry 26:1434–1442
Yarden, Y., Schlessinger, J. 1987b. Epidermal growth factor induces rapid, reversible aggregation of the purified epidermal growth factor receptor.Biochemistry 26:1443–1451
Zidovetzki, R., Yarden, Y., Schlessinger, J., Jovin, T.M. 1981. Rotational diffusion of epidermal growth factor complexed to cell surface receptors reflects rapid microaggregation and endocytosis of occupied receptors.Proc. Natl. Acad. Sci. USA 78:6981–6985
Zidovetzki, R., Yarden, Y., Schlessinger, J., Jovin, T.M. 1986. Microaggregation of hormone-occupied epidermal growth factor receptors on plasma membrane preparations.EMBO J. 5:247–250
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Azevedo, J.R., Johnson, D.A. Temperature-dependent lateral and transverse distribution of the epidermal growth factor receptor in A431 plasma membranes. J. Membrain Biol. 118, 215–224 (1990). https://doi.org/10.1007/BF01868605
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF01868605