Abstract
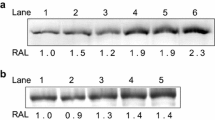
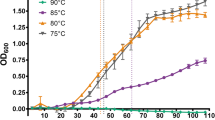
The whole-cell protein inventory of the deep-sea barophilic hyperthermophile Thermococcus barophilus was examined by one-dimensional SDS gradient gel electrophoresis when grown under different pressure conditions at 85°C (T opt). One protein (P60) with a molecular mass of approximately 60 kDa was prominent at low pressures (0.3 MPa hydrostatic pressure and 0.1 MPa atmospheric pressure) but not at deep-sea pressures (10, 30, and 40 MPa). About 17 amino acids were sequenced from the N-terminal end of the protein. Sequence homology analysis in the GenBank database showed that P60 most closely resembled heat-shock proteins in some sulfur-metabolizing Archaea. A high degree of amino acid identity (81%–93%) to thermosome subunits in Thermococcales strains was found. Another protein (P35) with molecular mass of approximately 35.5 kDa was induced at 40 MPa hydrostatic pressure but not under low-pressure conditions. No amino acid sequence homology was found for this protein when the 40 amino acids from the N-terminal end were compared with homologous regions of proteins from databases. A PTk diagram was generated for T. barophilus. The results suggest that P habitat is about 35 MPa, which corresponds to the in situ pressure where the strain was obtained.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received: May 14, 1999 / Accepted: July 30, 1999
Rights and permissions
About this article
Cite this article
Marteinsson, V., Reysenbach, AL., Birrien, JL. et al. A stress protein is induced in the deep-sea barophilic hyperthermophile Thermococcus barophilus when grown under atmospheric pressure. Extremophiles 3, 277–282 (1999). https://doi.org/10.1007/s007920050128
Issue Date:
DOI: https://doi.org/10.1007/s007920050128