Abstract
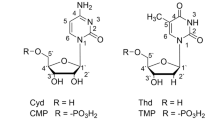
HENDRICKS1 has measured the interplanar cleavage spacings, d (001), of a number of organic salts of the clay mineral montmorillonite by means of nickel K? radiation. Among the cations studied were those of the purine bases guanine and adenine and the corresponding nucleosides guanosine and adenosine, which are 9-guanine d-ribofuranoside and 9-adenine d-ribofuranoside respectively2. By his measurements Hendricks showed that the arrangement of the atoms of the two purine ions is coplanar, thus confirming the view which is reached from consideration of molecular models. He also concluded that the nucleoside ions have a van der Waals 'thickness' between oxygen centres (of montmorillonite) only about 1.0 A. greater than would be required for a strictly coplanar atomic arrangement, and that the purine and sugar radicals of the nucleoside ions lie in two parallel planes about 1.5 A. apart, with the majority of the atoms lying probably in or near the two planes; one plane contains the purine radical, the other the ribose ring, and it is held that the hydroxyl groups as well as the primary alcohol group must be approximately in the plane of the purine radical, in order to accommodate this close lateral packing of atoms.
Similar content being viewed by others
Article PDF
References
J. Phys. Chem., 45, 65 (1941).
See Gulland, J. Chem. Soc., 1722 (1938).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
GULLAND, J., BARKER, G. & JORDAN, D. Configuration of Purine Nucleosides. Nature 151, 109 (1943). https://doi.org/10.1038/151109a0
Issue Date:
DOI: https://doi.org/10.1038/151109a0
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.