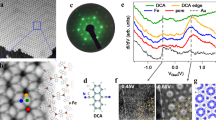
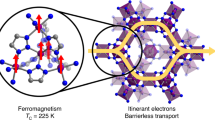
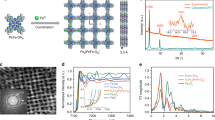
Abstract
THE ability of molecular materials to perform many of the optical, electronic and magnetic functions traditionally associated with extended two- and three-dimensional inorganic solids1,2 has given rise to intensive research on molecular electronics3,4. In the course of investigating the properties of a class of anionic metal clusters based on the vanadium oxide systems5–8, which bear analogy with those of bulk solid materials6, we have encountered unusual magnetic behaviour in a finite molecular system. A cluster containing 15 paramagnetic vanadium atoms consists of three distinct layers in each of which the magnetization shows a distinct temperature dependence. Analogous behaviour in bulk systems can be found in magnetic multilayers9 and also in copper oxide superconductors, where copper layers with strong antiferromagnetic coupling are separated by layers of rare-earth ions in which the coupling is very weak10. The behaviour of this cluster suggests the possibility of applications for molecular-scale switching.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Simon, J., Andre, J. J. & Skaulios, A. New J. Chem. 10, 295–311 (1986).
Magnetic Molecular Materials (eds Gatteschi, D., Kahn, O., Miller, J. S. & Palacio, F.) (Kluwer, Dordrecht, 1991).
Molecular Electronic Devices I: II (ed. Carter, F. L.) (Dekker, New York, 1982; 1987).
Zarembowitch, J. & Kahn, O. New J. Chem. 15, 181–190 (1991).
Pope, M. & Müller, A. Angew. Chem. Int. Ed. Engl. 30, 34–48 (1990).
Müller, A., Rohlfing, R., Penk, M. & Döring, J. Angew. Chem. Int. Ed Engl. 30, 588–590 (1991).
Müller, A. et al. Angew. Chem. Int. Ed. Engl. 30, (1991).
Müller, A., Döring, J. & Bögge, H. J. chem. Soc. Chem. Commun. 5, 273–274 (1991).
Shinjo, T. et al. J. magn. Magn. Mat. 54–57, 737–742 (1989).
Chemistry of the High Temperature Superconductors, Ch. 13; 14 (eds Nelson, D. L. & George, T. F.) (ACS Symposium Series 377, Washington DC, 1988).
Müller, A. & Döring, J. Angew. Chem. Int. Ed. Engl. 27, 1721 (1988).
Benelli, C., Dei, A., Gatteschi, D. & Pardi, L. Inorg. Chem. 27, 2831–2836 (1988).
Bencini, A. & Gatteschi, D. EPR of Exchange Coupled Systems (Springer, Berlin, 1990).
Bencini, A. & Gatteschi, D. in Transition Metal Chemistry, Vol. 8 (eds Melson, G. A. & Figgis, B. N.) (Dekker, New York, 1982).
Otake, M., Komiyama, Y. Otaki, T. J. phys. Chem. 77, 2896–2903 (1973).
Goodenough, J. B. Les Oxydes Des Métaux De Transition, 143; 297 (Gauthiers-Villars, Paris, 1973).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Gatteschi, D., Pardi, L., Barra, A. et al. Layered magnetic structure of a metal cluster ion. Nature 354, 463–465 (1991). https://doi.org/10.1038/354463a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/354463a0
This article is cited by
-
Synthesis, structures and applications of electron-rich polyoxometalates
Nature Reviews Chemistry (2018)
-
Tetranuclear dioxomolybdenum (VI) cluster anion, hydrogen bond interaction with 1,2-di(4-pyridyl)ethylene
Journal of Thermal Analysis and Calorimetry (2016)
-
Systematic Study of the Interaction Between VIV Centres and LnIII Ions in Well Defined {V 2 IV LnIII}{AsIIIW9O33}2 Sandwich-Type Clusters: Part 2
Journal of Cluster Science (2013)
-
Frustration signatures in the anisotropic model of a nine-spin s = 3/2 ring with bond defect
Journal of Nanoparticle Research (2011)
-
Lanthanide-Organic Frameworks Based on {Ln4(μ3-OH)4(μ2-OH)2} Cluster Units
Journal of Cluster Science (2010)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.