Abstract
The ability of glucagon to stimulate glycogen breakdown in liver played a key part in the classic identification of cyclic AMP and hormonally stimulated adenylate cyclase1. But several observations indicate that glucagon can exert effects independent of elevating intracellular cAMP concentrations2–7. These effects are probably mediated by an elevation8,9 of the intracellular concentration of free Ca2+ although the mechanism by which this occurs is unknown. We show here that glucagon, at the low concentrations found physiologically, causes both a breakdown of inositol phospholipids and the production of inositol phosphates. Indeed, we show that the glucagon analogue, (1-N-α-trinitrophenylhistidine,12-homo-arginine)glucagon (TH-glucagon), which does not activate adenylate cyclase or cause any increase in cAMP in hepatocytes yet can fully stimulate glycogenolysis, gluconeogenesis and urea synthesis10, stimulates the production of inositol phosphates. This stimulation of inositol phospholipid metabolism by low concentrations of glucagon provides a mechanism11,12 whereby glucagon can exert cAMP-independent actions on target cells. We suggest that hepatocytes possess two distinct receptors for glucagon, a GR-1 receptor coupled to stimulate inositol phospholipid breakdown and a GR-2 receptor coupled to stimulate adenylate cyclase activity.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Sutherland, E. W. & Rall, T. W. J. biol. Chem. 232, 1077–1091 (1958).
Birnbaum, M. J. & Fain, J. N. J. biol. Chem. 252, 528–535 (1977).
Okajima, F. & Ui, M. Archs Biochem. Biophys. 175, 549–557 (1976).
Cardenas-Tanus, R. & Garcia-Sainz, J. A. FEBS Lett. 143, 1–4 (1982).
Khan, B. A., Bregman, M. D., Nugent, C. A., Hruby, V. J. & Brendel, K. Biochem. biophys. Res. Commun. 93, 729–736 (1980).
Heyworth, C. M., Wallace, A. V. & Houslay, M. D. Biochem. J. 214, 99–110 (1983).
Heyworth, C. M. & Houslay, M. D. Biochem. J. 214, 93–98 (1983).
Sistaire, F. D., Picking, R. A. & Haynes, R. C. J. biol. Chem. 260, 12744–12747 (1985).
Mauger, J.-P. & Claret, M. FEBS Lett. 195, 106–110 (1986).
Corvera, S. et al. Biochim. biophys. Acta 804, 434–441 (1984).
Berridge, M. J. & Irvine, R. F. Nature 312, 315–321 (1984).
Downes, C. P. & Michell, R. H. Molec. Aspects Cell Regul. 4, 2–56 (1985).
Creba, J. A., Downes, C. P., Hawkins, P. T., Brewster, G., Michell, R. H. & Kirk, C. J. Biochem. J. 212, 733–747 (1983).
Houslay, M. D. & Elliott, K. R. F. FEBS Lett. 359–363 (1979).
Heyworth, C. M., Whetton, A. D., Wong, S., Martin, R. B. & Houslay, M. D. Biochem. J. 228, 593–603 (1985).
Heyworth, C. M., Hanski, E. & Houslay, M. D. Biochem. J. 222, 189–194 (1984).
Thomas, A. P., Alexander, J. & Williamson, J. F. J. biol Chem. 259, 5574–5584 (1984).
Bocckino, S. B., Blackmore, P. F. & Exton, J. H. J. biol Chem. 260, 14201–14207 (1985).
Heyworth, C. M., Wilson, S. R., Gawler, D. & Houslay, M. D. FEBS Lett. 187, 196–200 (1985).
Sonne, O., Berg, T. & Christofferson, T. J. biol. Chem. 253, 3203–3210 (1978).
Musso, G. F., Assoian, R. K., Kaiser, E. T., Kezdy, F. J. & Tager, H. S. Biochem. biophys. Res. Commun. 119, 713–719 (1984).
Heyworth, C. M. & Houslay, M. D. Biochem. J. 214, 547–552 (1983).
Berridge, M. J., Downes, C. P. & Hanley, M. R. Biochem. J. 206, 587–595 (1982).
Bregman, M. D., Trivedi, D. & Hruby, V. J. J. biol Chem. 255, 11725–11733 (1980).
Berridge, M. J., Dawson, R. M. C., Downes, C. P., Heslop, J. P. & Irvine, R. F. Biochem. J. 212, 473–482 (1983).
Houslay, M. D., Metcalfe, J. C., Warren, G. B., Hesketh, T. R. & Smith, G. A. Biochim. biophys. Acta 436, 489–494 (1976).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Wakelam, M., Murphy, G., Hruby, V. et al. Activation of two signal-transduction systems in hepatocytes by glucagon. Nature 323, 68–71 (1986). https://doi.org/10.1038/323068a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/323068a0
This article is cited by
-
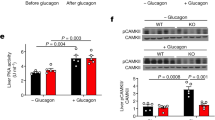
CD38/ADP-ribose/TRPM2-mediated nuclear Ca2+ signaling is essential for hepatic gluconeogenesis in fasting and diabetes
Experimental & Molecular Medicine (2023)
-
CD38-mediated Ca2+ signaling contributes to glucagon-induced hepatic gluconeogenesis
Scientific Reports (2015)
-
Current Insights and New Perspectives on the Roles of Hyperglucagonemia in Non-Insulin–Dependent Type 2 Diabetes
Current Hypertension Reports (2013)
-
Inositol-1,4,5-trisphosphate receptor regulates hepatic gluconeogenesis in fasting and diabetes
Nature (2012)
-
PKC stimulated by glucagon decreases UT-A1 urea transporter expression in rat IMCD
Pflügers Archiv - European Journal of Physiology (2008)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.