Abstract
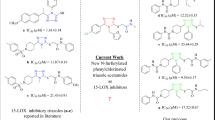
Methoxyalkyl thiazoles are novel 5-lipoxygenase inhibitors which are neither redox agents nor iron chelators and are exemplified by ICI211965 [1-(3-naphth-2-ylmethoxy)phenyl)-1-(thiazol-2-yl)propyl methyl ether]. ICI211965 potently inhibits LTC4 synthesis in murine macrophages (IC50=0.0085 μM) and its selectivity with respect to cyclo-oxygenase (>5800) is greater than any previously reported lipoxygenase inhibitor. ICI211965 also selectively inhibits LTB4 synthesis by human bloodin vitro (IC50=0.45 μM) and rat bloodex vivo (ED50=10 mg/Kg, p.o.). Methoxyalkyl thiazoles exhibit a tight structure activity relationship and resolution of a chiral member of the series demonstrates that 5-lipoxygenase inhibition resides largely in one enantiomer. Methoxyalkyl thiazoles represent the first class of agents for which 5-lipoxygenase inhibition is mediated by specific, enantioselective interaction with the enzyme.
Similar content being viewed by others
References
R. M. McMillan and S. J. Foster,Leukotriene B 4 and inflammatory disease. Agents and Actions,24, 114–119 (1988).
M. O'Donnell and A. Welton,Comparison of the pulmonary pharmacology of leukotrienes and PAF: effects of their antagonists. InTherapeutic approaches to inflammatory diseases (Eds. A. J. Lewis, N. S. Doherty and N. R. Ackerman) pp. 169–193, Elsevier, New York (1989).
J. A. Salmon, W. P. Jackson and L. G. Garland,Development of 5-lipoxygenase inhibitors: potential drugs for asthma and inflammation. InTherapeutic approaches to inflammatory diseases (Eds. A. J. Lewis, N. S. Doherty and N. R. Ackerman) pp. 137–146, Elsevier, New York (1989).
R. M. McMillan, D. J. Masters, V. C. Vickers, M. P. Dicken and V. N. Jacobs,Metabolism of unsaturated fatty acids by RBL-1 5-lipoxygenase: influence of substrate solubility and product inactivation. Biochim. Biophys. Acta,1005, 170–176 (1989).
S. J. Foster, P. Bruneau, E. R. H. Walker and R. M. McMillan,2-substituted indazolinones: orally active and selective 5-lipoxygenase inhibitors with anti-inflammatory activity. Br. J. Pharmacol.99, 113–118 (1990).
S. M. Coutts, A. Khandwala, R. van Inwegen, U. Chakraborty, J. Musser, J. Bruens, N. Jariwala, V. Dally-Meade, R. Ingram, T. Pruss, H. Jones, E. Neiss and I. Weinryb,Arylmethyl phenyl ethes. A new class of specific inhibitors of 5-lipoxygenase. InProstaglandins, leukotrienes and lipoxins (Ed. J. M. Bailey) pp. 627–637. Plenum Press, New York (1985).
R. M. McMillan, J.-M. Girodeau and S. J. Foster,Selective chiral inhibitors of 5-lipoxygenase with anti-inflammatory activity. Br. J. Pharmacol.101, 501–503 (1990).
D. Aharony, R. L. Stein, D. G. Redkar-Brown, S. J. Hubbs, E. Kusner and R. D. Krell,The mechanism of leukotriene inhibition of Rev-5901. Fed. Proc.,45, 659 (1986).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
McMillan, R.M., Bird, T.G.C., Crawley, G.C. et al. Methoxyalkyl thiazoles: A novel series of potent, orally active and enantioselective inhibitors of 5-lipoxygenase. Agents and Actions 34, 110–112 (1991). https://doi.org/10.1007/BF01993252
Issue Date:
DOI: https://doi.org/10.1007/BF01993252