Summary
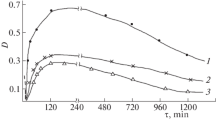
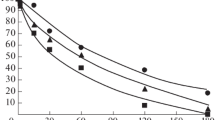
Interaction of seryl trihydroxy-benzyl-hydrazine with a highly purified preparation of Dopa decarboxylase from pig kidney has been studied. This compound was found not to be a powerful inhibitor in vitro. Kinetic and spectral data suggest some possibilities on the binding nature of the inhibitor and substrates.
Similar content being viewed by others
References
W. P. Burkard, K. F. Gey and A. Pletscher, Experientia15, 411 (1962).
W. P. Burkard, K. F. Gey and A. Pletscher, Arch. Biochem. Biophys.107, 187 (1964).
C. Borri Voltattorni, A. Minelli and C. Turano, Riun. Naz. Soc. ital. Biochim. Riass. Comm. No. 48 (1970).
J. G. Christenson, W. Dairman and S. Udenfriend, Arch. Biochem. Biophys.141, 356 (1970).
A. F. Sherald, J. C. Sparrow and T. R. F. Wright, Anal. Biochem.56, 300 (1973).
A. Charteris and R. John, Ann. Biochem.66, 365 (1975).
C. Borri-Voltattorni, A. Minelli and C. Turano, FEBS Letters17, 231 (1971).
E. Barboni, C. Borri-Voltattorni, A. Rosei, A. Fiori, A. Minelli and C. Turano, Comm. submitted to 2nd Italian Biochem. Congress, Venezia (1976).
R. M. Krupka and K. J. Laidler, J. Am. chem. Soc.83, 1445 (1961).
A. Fiori, C. Turano, C. Borri-Voltattorni, A. Minelli and M. Codini, FEBS Letters54, 122 (1975).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
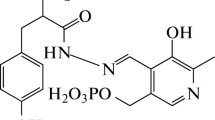
Borri-Voltattorni, C., Minelli, A. & Borri, P. Interaction of N-(DL-seryl)N′-(2,3,4-trihydroxybenzyl)-hydrazine with L-Dopa decarboxylase from pig kidney. Experientia 33, 158–160 (1977). https://doi.org/10.1007/BF02124039
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF02124039