Abstract
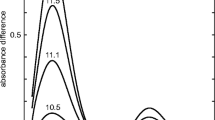
It was shown by means of IR, UV, and PMR spectra that β-(3-nitro-2-pyridyl)pyruvic acid esters are practically completely enolized in the crystal state and in solution; ethyl β-(3-nitro-4-pyridyl)pyruvate has an enol structure in the crystalline state and in pyridine solution but exists as a mixture of keto and enol forms in low-polarity solvents.
Similar content being viewed by others
Literature cited
M. Becker, Ber. Bunsenges. Phys. Chem., 68, 669 (1964).
G. W. Wheland, in: Advances in Organic Chemistry, Wiley (1960).
M. Nishio and T. Ito, Agric. Biol. Chem., 29, 1119 (1965).
B. Frydman, S. J. Reil, J. Boned, and H. Rapoport, J. Org. Chem., 33, 3762 (1968).
B. Frydman, M. E. Despuy, and H. Rapoport, J. Am. Chem. Soc., 87, 3530 (1965).
L. Bellamy, Infrared Spectra of Complex Molecules, Methuen (1958).
L. N. Yakhontov and V. A. Azimov, Tetrahedron Lett., 1909 (1969).
M. H. Fischer and A. R. Matzuk, J. Heterocycl. Chem., 6, 775 (1969).
Author information
Authors and Affiliations
Additional information
Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 3, pp. 389–393, March, 1974.
Rights and permissions
About this article
Cite this article
Peresleni, E.M., Uritskaya, M.Y., Azimov, V.A. et al. Structure, tautomerism, and transformations of β-(3-nitro-2-pyridyl)- and β-(3-nitro-4-pyridyl)pyruvic acid esters. Chem Heterocycl Compd 10, 340–344 (1974). https://doi.org/10.1007/BF00472426
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00472426