Abstract
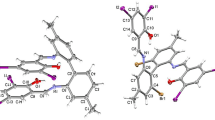
The molecular structure of phenol-pendant cyclam-zinc(II) complex,4a, has been determined by X-ray structure analysis. Crystals of4a · ClO4 · CH3OH (C16H27N4OZn · ClO4 · CH3OH) are monoclinic, space groupP21/nn, with four molecules in the unit cell of dimensionsa=31.198(2) Å,b=8.426(1) Å,c=8.214(1) Å, andβ=93.96(1)°. The structure was solved by the heavy atom method and refined anisotropically toR=0.044,R w=0.062 for 1551 independent reflections. The complex assumes a five-coordinate, square pyramidal geometry, where zinc(II) is surrounded by the cyclam moiety in a planar fashion with the pendant phenolate anion occupying an axial position. An extremely short Zn-O(phenolate) bond distance of 1.983(5) Å, in conjunction with the 0.288 Å displacement of Zn(II) above the cyclam N4 plane toward the phenolate, accounts for the extremely low pK a value of 5.8 for the pendant phenol. These facts about4a, in comparison with the previous findings for the Ni(II) (4b) and Cu(II) complexes (4c) with the same ligand, illustrate well the characteristics of zinc(II) ion coordination properties.
Similar content being viewed by others
References
P. Woolley:Nature (London) 258, 677 (1975). J. T. Groves, and R. R. Chambers, Jr:J. Am. Chem. Soc. 106, 630 (1984).
E. Kimura, T. Shiota, T. Koike, M. Shiro, and M. Kodama:J. Am. Chem. Soc. 112, 5805 (1990).
E. Kimura, T. Koike, and K. Toriumi:Inorg. Chem. 27, 3687 (1988).
Y. Pocker and S. Sarkanen:Adv. Enzymol. 47, 149 (1987). Y. Pocker, J. T. Stone:J. Am. Chem. Soc. 87, 5497 (1965).
M. Kodama and E. Kimura:J. Chem. Soc., Dalton Trans. 2269 (1977).
T. Ito, M. Kato, and H. Ito:Bull. Chem. Soc. Jpn. 57, 2634 (1984). M. Kato and T. Ito:Inorg. Chem. 24, 509 (1985).
Y. Iitaka, T. Koike, and E. Kimura:Inorg. Chem. 25, 402 (1986).
E. Kimura, T. Koike, K. Uenishi, M. Hediger, M. Kuramoto, S. Joko, Y. Arai, M. Kodama, and Y. Iitaka:Inorg. Chem. 26, 2975 (1987).
E. Kimura, T. Koike, and M. Takahashi:J. Chem. Soc., Chem. Commun. 385 (1985).
E. Kimura, M. Yamaoka, M. Morioka, and T. Koike:Inorg. Chem. 25, 3883 (1986).
E. Kimura, T. Koike, H. Nada, and Y. Iitaka:Inorg. Chem. 27, 1036 (1988).
V. J. Thőm, C. C. Fox, J. C. A. Boeyens, and R. D. Hancock:J. Am. Chem. Soc. 106, 5947 (1984).
Our recent papers about Zn(II) chemistry: E. Kimura, T. Koike, T. Shiota, and Y. Iitaka:Inorg. Chem. 29, 1460 (1990). E. Kimura, and T. Koike:Comments on Inorg. Chem. Gordon and Breach Science Publishers: New York, Vol. 11, 285 (1991).
Author information
Authors and Affiliations
Additional information
This paper is dedicated to the memory of the late Dr C. J. Pedersen.
Rights and permissions
About this article
Cite this article
Kimura, E., Kurosaki, H., Koike, T. et al. X-ray structural study of a zinc(II) inclusion complex of a phenolate-pendant cyclam. J Incl Phenom Macrocycl Chem 12, 377–387 (1992). https://doi.org/10.1007/BF01053875
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01053875