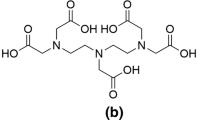
Abstract
Cyclodextrins form complexes with lanthanide ions in basic aqueous solutions. This complex formation in basic solution dramatically enhances the solubility of lanthanide ions, which are otherwise insoluble due to the formation of hydroxide gels. Solutions of the γ-cyclodextrin-Ce3+ complex effectively hydrolyze 2′-deoxyadenosine-5′-monophosphate to 2′-deoxyadenosine.
Similar content being viewed by others
Notes and References
Y. Matsumoto and M. Komiyama:Chem. Express 7, 785 (1992).
M. Komiyama, K. Matsumura, K. Yonezawa, and Y. Matsumoto:Chem. Express 8, 85 (1993).
M. Komiyama, M. Yashiro, Y. Matsumoto, J. Sumaoka, and K. Matsumura:Nippon Kagaku Kaishi, 411 (1993).
M. Komiyama, Y. Matsumoto, N. Hayashi, K. Matsumura, N. Takeda, and K. Watanabe:Polym. J. 25, 1211 (1993).
T. Shiiba, K. Yonezawa, N. Takeda, Y. Matsumoto, M. Yashiro, and M. Komiyama:J. Mol. Catal. 84, L21 (1993).
M. Komiyama, T. Shiiba, Y. Takahashi, N. Takeda, K. Matsumura, and T. Kodama:Supramol. Chem. in press.
M. Komiyama, T. Shiiba, Y. Takahashi, N. Takeda, K. Matsumura, T. Kodama, and M. Yashiro:Nucleosides and Nucleotides in press.
B.K. Takasaki and J. Chin:J. Am. Chem. Soc. 116, 1121 (1994).
M. Komiyama, T. Kodama, N. Takeda, J. Sumaoka, T. Shiiba, Y. Matsumoto, and M. Yashiro:J. Biochem. 115, 809 (1994).
M. Komiyama, T. Shiiba, Y. Takahashi, N. Takeda, H. Uchida, J. Sumaoka, T. Kodama, and M. Yashiro: submitted.
M. Komiyama, K. Matsumura, and Y. Matsumoto:J. Chem. Soc. Chem. Commun., 640 (1992).
N. Hayashi, N. Takeda, T. Shiiba, M. Yashiro, K. Watanabe, and M. Komiyama:Inorg. Chem. 32, 5899 (1993).
J.R. Morrow, V.M. Shelton, L.A. Buttrey, and K.A. Berback:J. Am. Chem. Soc. 114 1903 (1992).
J.R. Morrow and K.A. Kolasa:Inorg. Chem. 32, 3983 (1983).
J. Ciesiolka, T. Marciniec, and W.J. Krzyzosiak:Eur. J. Biochem. 182, 445 (1989).
K. Matsumura and M. Komiyama:J. Inorg. Biochem. in press.
J. Sumaoka, M. Yashiro, and M. Komiyama:J. Chem. Soc., Chem. Commun., 1707 (1992).
C.V. Banks and D.W. Klingman:Anal. Chim. Acta 15, 356 (1956).
M.L. Bender and M. Komiyama:Cyclodextrin Chemistry, Springer-Verlag: New York, 1977; F. Vögtle:Supramolecular Chemistry, Wiley: New York, 1991.
The diameter of the hydrated Pr3+ ion is estimated by the simple summation of the Pr3+−O distances (A. Habenschuss and F.H. Spedding:J. Chem. Phys. 70, 3758 (1979)) and the diameter of the water molecule.
Such a type of coordination of metal ions with CD has been anticipated in a recent review (J. Szejtli:Stärke 42, 444 (1990)).
The solubilization of lanthanide ions by cyclodextrins is commonly observed even at pH from 8 to 11. Therefore, the proton dissociation of the hydroxyl groups (pK a ca. 12) is not necessary for the solubilization of lanthanide ions, in contrast to the solubilization of Cu2+ by CDs (Y. Matsui, T. Kurita, M. Yagi, T. Okayama, K. Mochida and Y. Date:Bull. Chem. Soc. Jpn. 48, 2187 (1975)).
H. Grasdalen, T. Anthonsen, O. Harbitz, B. Larsen, and O. Smidsrød:Acta Chem. Scand. A 32, 31 (1978); S.J. Angyal and D. Greeves:Aust. J. Chem. 29, 1223 (1976).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Yashiro, M., Miyama, S., Takarada, T. et al. Solubilization of lanthanide ions by cyclodextrins in basic aqueous solutions. J Incl Phenom Macrocycl Chem 17, 393–397 (1994). https://doi.org/10.1007/BF00707134
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00707134