Abstract
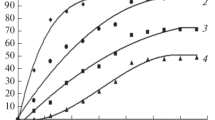
The dissolution behaviour of two alloys, which represent the metallic precipitates, occurring in the dissolution step of a nuclear reprocessing plant was investigated. The solubility of these alloys, containing Mo, Ru, Rh and Pd, in HNO3-solutions is rather low and dissolution ceases after some time, caused by passivation. A significant acceleration of the dissolution rate takes place if ozone is passed through the alloy/HNO3-suspension, or if an electrolytic dissolver with or without an oxidation catalyst is used. Moreover, Ru can be separated as RuO4 by distillation.
Similar content being viewed by others
References
F. J. SMITH, M. F. MCDUFFIE, Sep. Sci. Technol., 16 (1981) 1071.
K. NAITO, T. MATSUI, Y. TANAKA, J. Nucl. Sci. Technol., 23 (1986) 540.
H. KLEYKAMP, Kinetics of the Dissolution of metallic fission product precipitates in nitric acid, Proc. Annual Meating on Nuclear Technology, Munich, FRG, 1985, p. 329.
H. KLEYKAMP, Nucl. Technol., 80 (1988) 412.
H. T. BAKER, P. E. BROWN, R. J. PATEMAN, K. L. WILKINSON, Report AERE-G 3408, Atomic Energy Research Establishment, Harwell, 1985.
J. O. PASCHOAL, H. KLEYKAMP, F. THÜMMLER, Z. Metallkunde, 74 (1983) 652.
G. BAUMGÄRTEL, Kernforschungszentrum Karlsruhe, FRG, personal communication.
G. MILAZZO, S. CAROLI, Tables of Standard Electrode Potentials, John Wiley & Sons, New York, 1978.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Geckeis, H., Neumann, W. & Müller, W. Dissolution of platinum metal alloys contained in the feed clarification sludge. Journal of Radioanalytical and Nuclear Chemistry, Articles 152, 199–206 (1991). https://doi.org/10.1007/BF02042153
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02042153