Abstract
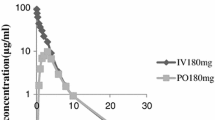
The pharmacokinetics of primaquine was studied in calves of 180–300 kg live weight. Primaquine was injected at 0.29 mg/kg (0.51 mg/kg as primaquine diphosphate) intravenously (IV) or subcutaneously (SC) and the plasma concentrations of primaquine and its metabolite carboxyprimaquine were determined by high-performance liquid chromatography. The extrapolated concentration of primaquine at zero time after IV administration was 0.50±0.48 µg/ml (mean ±SD) which decreased with an elimination half-life of 0.16±0.07 h. Primaquine was rapidly converted to carboxyprimaquine after either route of administration. The peak concentration of carboxyprimaquine was 0.50±0.08 µg/ml at 1.67±0.15 h after IV administration. The corresponding value was 0.47±0.07 µg/ml at 5.05±1.20 h after SC administration. The elimination half-lives of carboxyprimaquine after IV and SC administration were 15.06±0.99 and 12.26±3.06 h, respectively. The areas under the concentration-time curve for carboxyprimaquine were similar following either IV or SC administration of primaquine; the values were 11.85±2.62 µg.h/ml after the former and 10.95±2.65 µg.h/ml after the latter. The mean area under the concentration-time curve for primaquine was less than 0.1 µg.h/ml after either route of administration.
Similar content being viewed by others
Abbreviations
- AUC:
-
area under the concentration-time curve
- CPRQ:
-
carboxyprimaquine
- IV:
-
intravenous
- 6M8AQ:
-
6-methoxy-8-aminoquinoline
- PRQ:
-
primaquine
- SC:
-
subcutaneous
References
Baker, J.K., McChesney, J.D., Hufford, C.D. and Clark, A.M., 1982. High-performance liquid chromatographic analysis of the metabolism of primaquine and the identification of a new mammalian metabolite.Journal of Chromatography,230, 69–77
Baker, J.K., Bedford, J.A., Clark, A.M. and McChesney, J.D., 1984. Metabolism and distribution of primaquine in monkeys.Pharmaceutical Research,1, 98–100
Bates, M.D., Meshnick, S.R., Sigler, C.I., Leland, P. and Hollingdale, M.R., 1990. In vitro effects of primaquine and primaquine metabolites on exoerythrocytic stages ofPlasmodium berghei.American Journal of Tropical Medicine and Hygiene,42, 532–537
Baty, J.D., Price-Evans, D.A. and Robinson, P.A., 1975. The identification of 6-methyoxy-8-aminoquinoline as a metabolite of primaquine in man.Mass Spectrometry,2, 304–306
Brossi, A., Millet, P., Landau, I., Bembenek, M.E. and Abell, C.W., 1987. Antimalarial activity and inhibition of monoamine oxidases A and B by exoerythrocytic antimalarials.FEBS Letters,214, 291–294
Clark, A.M., Baker, J.K. and McChesney, J.D., 1984. Excretion, distribution and metabolism of primaquine in rats.Journal of Pharmaceutical Sciences,73, 502–506
Endoh, Y.S., Yoshimura, H., Sasaki, N., Ishihara, Y., Sasaki, H., Nakamura, S., Inoue, Y. and Nishikawa, M., 1992. High-performance liquid chromatographic determination of pamaquin, primaquine and carboxyprimaquine in calf plasma using electrochemical detection.Journal of Chromatography,579, 123–129
Lee, C.C., Kinter, L.D. and Heiffer, M.H., 1981. Subacute toxicity of primaquine in dogs, monkeys and rats.Bulletin of the World Health Organization,59, 439–448
Link, C.M., Theoharides, A.D., Anders, J.C., Chung, H. and Canfield, C.J., 1985. Structure-activity relationships of putative primaquine metabolites causing methemoglobin formation in canine hemolysates.Toxicology and Applied Pharmacology,81, 192–202
Mihaly, G.W., Ward, S.A., Edwards, G., Orme, M.L'E. and Breckenridge, A.M., 1984. Pharmacokinetics of primaquine in man: Identification of the carboxylic acid derivative as a major plasma metabolite.British Journal of Clinical Pharmacology,17, 441–446
Mihaly, G.W., Ward, S.A., Edwards, G., Nicholl, D.D., Orme, M. L'E. and Breckenridge, A.M., 1985. Pharmacokinetics of primaquine in man. I. Studies of the absolute bioavailability and effects of dose size.British Journal of Clinical Pharmacology,19, 745–750
Nova, M.V., Parkhurst, G.W., Thomas, R.W. and Carson, P.E., 1984. High-performance liquid chromatographic-electrochemical assay method for primaquine in plasma and urine.Journal of Chromatography,307, 451–456
Parkhurst, G.W., Nova, M.V., Thomas, R.W. and Carson, P.E., 1984. High-performance liquid chromatographic-ultraviolet determination of primaquine and its metabolites in human plasma and urine.Journal of Pharmaceutical Sciences,73, 1329–1331
Schmidt, I.G. and Schmidt, L.H., 1951. Neurotoxicity of the 8-aminoquinolines. III. The effect of pentaquine, isopentaquine, primaquine and pamaquine on the central nervous system of rhesus monkeys.Journal of Neuropathology and Experimental Neurology,10, 231–256
Ward, S.A., Mihaly, G.W., Edwards, G., Looareesuwan, S., Phillips, R.E., Chanthavanich, P., Warrell, D.A., Orme, M. L'E. and Breckenridge, A.M., 1985. Pharmacokinetics of primaquine in man. II. Comparison of acute vs chronic dosage in Thai subjects.British Journal of Clinical Pharmacology,19, 751–755
Yamaoka, K., 1986.Methods for Pharmacokinetic Analysis by Personal Computer, (Nanko-do, Tokyo)
Zhang, Z.H., 1987. Elimination of the gametocytes ofTheileria annulata of cattle by primaquine phosphate.Veterinary Pharmacology,23, 11–21
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Yoshimura, H., Endoh, Y.S., Ishihara, Y. et al. The pharmacokinetics of primaquine in calves after subcutaneous and intravenous administration. Vet Res Commun 17, 129–136 (1993). https://doi.org/10.1007/BF01839241
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01839241