Abstract
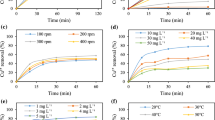
The adsorption technique using wollastonite has been applied for the removal of Cu(II) from aqueous solutions. The low concentration, high temperature and alkaline pH favor the removal of Cu(II). The Langmuir isotherm was used to represent the equilibrium data at different temperatures. The apparent heat of adsorption has been found to be 5.926 Cal mol−1. The uptake of Cu(II) is diffusion controlled and the mass transfer coefficient is 3.6 × 10−5 cm s−1. The maximum removal of Cu(II) in alkaline medium has been explained on the basis of the uptake of hydrolyzed adsorbate species by the active surface sites of adsorbent.
Similar content being viewed by others
References
Am. Public Health Assoc.: 1980,Standard Methods for the Examination of Water and Wastewater, 15th ed., APHA-WPCF-AWWA, Washington, D.C.
Bye, G. C., McEvoy, M., and Malati, M. A.: 1982,J. Chem. Tech. Biotechnol. 32, 781.
Choudhury, G. R. and Vaidya, S. H.: 1982,J. Chem. Tech. Biotechnol. 32, 883.
Crank, J.: 1965, The Mathematics of Diffusion, Clarendon Press, London.
Dean, J. G., Borqui, F. L., and Labouette, K. H.: 1972,Environ. Sci. Tech. 6, 518.
Elliot, H. A. and Huang, C. P.: 1981,Water Res. 15, 849.
Gupta, G. C. and Harrison, F. L.: 1980,Water, Air, and Soil Pollut. 13, 247.
Gupta, G. C. and Harrison, F. L.: 1981,Water, Air, and Soil Pollut. 15, 323.
Gupta, G. C. and Harrison, F. L.: 1982,Water, Air, and Soil Pollut. 17, 357.
Gupta, G. C. and Harrison, F. L.: 1983,Pollut. Res. 1, 1.
Guy, R. D., Chakrabarti, C. L., and Schramm, L. L.: 1975,Can. J. Chem. 53, 661.
Huang, C. P. and Wirth, P. K.: 1982,J. Env. Eng. Div. Am. Soc. Civ. Eng. 108, 1280.
Huang, C. P. and Blankenship, D. W.: 1984,Water Res. 18, 37.
Indian Standard Methods of Chemical Analysis of Fire clay and Silica Refractory Materials: 1960, Is: 1527.
Jain, K. K., Singh, V. N., and Prasad, G.: 1982,Proc. 7th International Clay Conf. 137.
Kishk, F. M. and Hassan, M. N.: 1973,Plant & Soil 39, 497.
Lagergren, S.: 1898,Bil. K. Svenska Vetenskapsakad. Hand. 24.
McConnel, B. L., Williams, K. C., Daniel, J. L., Stanton, J. H., Irby, B. N., Dugger, D. L., and Maatman, R. W.: 1964,J. Phys. Chem. 68, 2941.
McLaren, R. G. and Crawford, D. V.: 1973,J. Soil Sci. 24, 443.
McKay, G., Otterburn, M. S., and Sweeney, A. G.: 1981,Water Res. 15, 327.
Newton, D. W., Ellis, R. Jr., and Paulsen, G. M.: 1976,J. Env. Qual. 5, 251.
Panday, K. K, Prasad, G., and Singh, V. N.: 1984a,Water Res. (accepted).
Panday, K. K, Prasad, G., and Singh, V. N.: 1984b,Indian J. Tech. 22, 70.
Poots, V. J. P., McKay, G., and Healy, J. J.: 1978,J. Wat. Poll. Contr. Fed. 50, 926.
Stumm, W. and O'Melia, C. R.: 1968,J. Am. Wat. Wks. Ass. 60, 514.
Tallmange, J. A.: 1965, “Nonferrous Metals”, in Gurham, C. F. (ed.), Chemical Technology, Vol. 2, Academic Press Inc., New York.
Tewari, P. H., Campbell, A. R., and Lee, W.: 1972,Can. J. Chem. 50, 1642.
Weber, W. J. Jr. and Morris, J. C.: 1963,J. San. Eng. Div. Am. Soc. Civ. Eng. 89, SA2, 31.
Weber, W. J. Jr. and Keinath, T. M.: 1967,Chem. Eng. Prog. Symp. Ser. 63, 79.
Zazic, J. E.: 1971, Water and its Impurities, Vol. 1, Marcel Dekker Inc., New York.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Panday, K.K., Prasad, G. & Singh, V.N. Use of wollastonite for the treatment of Cu(II) rich effluents. Water Air Soil Pollut 27, 287–296 (1986). https://doi.org/10.1007/BF00649410
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00649410