Abstract
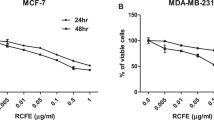
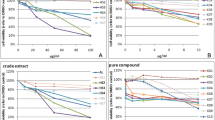
Indole-3-carbinol (I3C) is a promising phytochemical agent in chemoprevention of breast cancer. Our present study is the first description of I3C that significantly inhibits the cell adhesion, spreading and invasion associated with an up-regulation of PTEN (a tumor suppressor gene) and E-cadherin (a regulator of cell–cell adhesion) expression in T47-D human breast cancer cells. Therefore, I3C exhibits anti-cancer activities by suppressing breast tumor cell growth and metastatic spread. Metastatic breast cancer is a devastating problem, clinical application of I3C as a potent chemopreventive agent may be helpful in limiting breast cancer invasion and metastasis.
Similar content being viewed by others
References
Liu H, Wormke M, Safe SH, Bjeldanes LF: Indolo[3,2-b]carbazole: a dietary-derived factor that exhibits both antiestrogenic and estrogenic activity. J Natl Cancer Inst 86: 1758–1765, 1994
Tiwari RK, Guo L, Bradlow HL, Telang NT, Osborne MP: Selective responsiveness of human breast cancer cells to indole-3-carbinol, a chemopreventive agent. J Natl Cancer Inst 86: 126–131, 1994
Wong GY, Bradlow L, Sepkovic D, Mehl S, Mailman J, Osborne MP: Dose-ranging study of indole-3-carbinol for breast cancer prevention. J Cell Biochem 28–29: 111–116, 1997
Chen I, Safe S, Bjeldanes L: Indole-3-carbinol and diindolylmethane as aryl hydrocarbon (Ah) receptor agonists and antagonists in T47D human breast cancer cells. Biochem Pharmacol 51: 1069–1076, 1999
Cover CM, Hsieh SJ, Cram EJ, Hong C, Riby JE, Bjeldanes LF, Firestone GL: Indole-3-carbinol and tamoxifen cooperate to arrest the cell cycle of MCF-7 human breast cancer cells. Cancer Res 59: 1244–1251, 1999
Carughi A: Antiproliferative effect of a blend of extracts from cruciferous vegetables on human breast cancer. FASEB J 11: A369, 1997
Bose S, Wang SI, Terry MB, Hibshoosh H, Parsons R: Allelic loss of chromosome 10q23 is associated with tumor progression in breast carcinomas. Oncogene 17: 123–127, 1998
Hahn M, Wieland I, Koufaki ON, Gorgens H, Sobottka SB, Schackert G, Schackert HK: Genetic alterations of the tumor suppressor gene PTEN/MMAC1 in human brain metastases. Clin Cancer Res 5: 2431–2437, 1999
Fan S, Wang JA, Yuan RQ, Ma YX, Meng Q, Erdos MR, Brody LC, Goldberg ID, Rosen EM: BRCA1 as a potential human prostate tumor suppressor: modulation of proliferation, damage responses and expression of cell regulatory proteins. Oncogene 16: 3069–3082, 1998
Li J, Yen C, Liaw D, Podsypanina K, Bose S, Wang SI, Puc J, Miliaresis C, Rodgers L, McCombie R, Bigner SH, Giovanella BC, Ittmann M, Tycko B, Hibshoosh H, Wigler MH, Parsons R: PTEN, a putative protein tyrosine phosphatase gene mutated in human brain, breast, and prostate cancer. Science 275: 1943–1947, 1997
Tamura M, Gu J, Matsumoto K, Aota S, Parsons R, Yamada KM: Inhibition of cell migration, spreading, and focal adhesions by tumor suppressor PTEN. Science 280: 1614–1617, 1998
Tamura M, Gu J, Takino T, Yamada KM: Tumor suppressor PTEN inhibition of cell invasion, migration, and growth: differential involvement of focal adhesion kinase and p130Cas. Cancer Res 59: 442–449, 1999
Tamura M, Gu J, Danen EH, Takino T, Miyamoto S, Yamada KM: PTEN interactions with focal adhesion kinase and suppression of the extracellular matrix-dependent phosphatidylinositol 3-kinase/Akt cell survival pathway. J Biol Chem 274: 20693–20703, 1999
Clark EA, Brugge JS: Integrins and signal transduction pathways: the road taken. Science 268: 233–239, 1995
Whang YE, Wu X, Suzuki H, Reiter RE, Tran C, Vessella RL, Said JW, Isaacs WB, Sawyers CL: Inactivation of the tumor suppressor PTEN/MMAC1 in advanced human prostate cancer through loss of expression. Proc Natl Acad Sci USA 95: 5246–5250, 1998
Hahn M, Wieland I, Koufaki ON, Gorgens H, Sobottka SB, Schackert G, Schackert HK: Genetic alterations of the tumor suppressor gene PTEN/MMAC1 in human brain metastases. Clin Cancer Res 5: 2431–2437
Bracke ME, Van Roy FM, Mareel MM: The E-cadherin/ catenin complex in invasion and metastasis. Curr Top Microbiol Immunol 213: 123–161, 1996
Siitonen SM, Kononen JT, Helin HJ, Rantala IS, Holli KA, Isola JJ: Reduced E-cadherin expression is associated with invasiveness and unfavorable prognosis in breast cancer. Am J Clin Pathol 105: 394–402, 1996
Berx G, Cleton-Jansen AM, Strumane K, de Leeuw WJ, Nollet F, van Roy F, Cornelisse C: E-cadherin is inactivated in a majority of invasive human lobular breast cancers by truncation mutations throughout its extracellular domain. Oncogene 13: 1919–1925, 1996
Handschuh G, Candidus S, Luber B, Reich U, Schott C, Oswald S, Becke H, Hutzler P, Birchmeier W, Hofler H, Becker KF: Tumour-associated E-cadherin mutations alter cellular morphology, decrease cellular adhesion and increase cellular motility. Oncogene 18: 4301–4312, 1999
Mbalaviele G, Dunstan CR, Sasaki A, Williams PJ, Mundy GR, Yoneda T: E-cadherin expression in human breast cancer cells suppresses the development of osteolytic bone metastases in an experimental metastasis model. Cancer Res 56: 4063–4070, 1996
Bracke ME, Charlier C, Bruyneel EA, Labit C, Mareel MM, Castronovo V: Tamoxifen restores the E-cadherin function in human breast cancer MCF-7/6 cells and suppresses their invasive phenotype. Cancer Res 54: 4607–4609, 1994
Cos S, Fernandez R, Guezmes A, Sanchez-Barcelo EJ: Influence of melatonin on invasive and metastatic properties of MCF-7 human breast cancer cells. Cancer Res 58: 4383–4390, 1998
Rights and permissions
About this article
Cite this article
Meng, Q., Goldberg, I.D., Rosen, E.M. et al. Inhibitory effects of Indole-3-carbinol on invasion and migration in human breast cancer cells. Breast Cancer Res Treat 63, 147–152 (2000). https://doi.org/10.1023/A:1006495824158
Issue Date:
DOI: https://doi.org/10.1023/A:1006495824158