Abstract
ICRF-187 was given to 62 evaluable patients with advanced solid tumors in a Phase I clinical trial. Weekly infusions were given in dosages ranging from 0,85 g/m2 to 7.42 g/m2 for a total of four weeks with a two week rest period between courses. Dose-limiting hematological toxicity was seen in heavily pretreated patients at a dose of 3.8 g/m2/week. All patients also developed reversible SGOT elevations. In patients with less prior therapy hematologic toxicity was not dose-limiting but hepatotoxicity, manifest by transient SGOT levels greater than 5 times baseline was seen at 7.42 g/m2/week even though only 3/6 patients could receive 4 consecutive weekly doses. At virtually all dose levels tested some patients developed anemia. Other toxicities, including alopecia, nausea, vomiting and reversible serum amylase elevations, were mild.
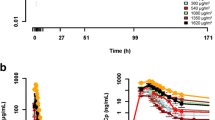
Cumulative monthly doses achieved on this weekly schedule are significantly higher than a 48-hour infusion or daily times 3 or 5 schedule in adults and a daily times 3 schedule in children. Pharmacokinetic studies in eight patients indicate that the drug disappears from the plasma biphasically with a terminal t1/2 of 3.2 +0.9 hr. The total clearance was 288.7 + 85.0 ml/hr/kg and the volume of distribution (Vda) was 1.3 ± 0.4 1/kg. Pharmacokinetics were not dose-dependent from 3.8–7.4 g/m2 and no difference in pharmacokinetics was found in patients studied during the first and second treatments of a course.
If Phase II trials of ICRF-187 are to be pursued on this schedule, appropriate doses would be 3.8 g/m2/week × 4 for heavily pretreated and 7.42 g/m2/week for “good risk” patients. Because of erratic hematologic toxicity in heavily pretreated patients, some might only tolerate three weekly doses. In good risk patients transaminitis was significant but reversible, thus, Phase II protocols should include dose escalation schemata.
Similar content being viewed by others
References
Flannery EP, Corder MP, Shehan TF, Pajak TF, Bateman JR: Phase II study of ICRF-159 in non-Hodgkin's lymphomas. Cancer Treat Rep 62:465–467, 1978
Olweny CLM, Masaba JP, Sikenunda W, Toya T: Treatment of Kaposi's sarcoma with ICRF-159 (NSC-129943). Cancer Treat Rep 60:111–113, 1976
Shah MK, Engstrom PF, Catalano RB, Paul AR, Bellet RE, Creech RH: Phase II trial of Razoxane (ICRF-159) in patients with squamous cell carcinoma of the head and neck previously exposed to systemic chemotherapy. Cancer Treat Rep 66:557–558, 1982
Bakowski MT, Prentice HG, Lister TA, Malpas JS, McElwain TJ, Powles RL: Limited activity of ICRF-159 in advanced acute leukemia. Cancer Treat Rep 63:127–129, 1979
Bakowski MT, Brearley RL, Wrigley HM: Treatment of blast cell crisis of chronic myeloid leukemia with ICRF-159 (Razoxane). Cancer Treat Rep 63:2085–2087, 1979
Creech RH, Engstrom PF, Harris D, Catalano RB, Bellet RE: Phase II study of ICRF-159 in refractory metastatic breast cancer. Cancer Treat Rep 63:111–114, 1979
Bellet RE, Engstrom PF, Catalano RB, Creech RH, Mastrangelo MJ: Phase II study of ICRF-159 in patients with metastatic colorectal carcinoma previously exposed to systemic chemotherapy. Cancer Treat Rep 60:1395–1397, 1976
Douglass HO Jr, MacIntyre JM, Kaufman J, vonHoff D, Engstrom PF, Klaassen D: Eastern Cooperative Oncology Group Phase II studies in advanced measurable colorectal cancer. I. Razoxane, Yoshi 64, piperazinedione, and Lomustine. Cancer Treat Rep 69:543–545, 1985
Paul AR, Catalano RB, Engstrom PF: Phase III study of ICRF-159 vs. 5-FU in the treatment of advanced metastatic colorectal carcinoma. Cancer Treat Rep 64:1047–1049, 1980
Chiuten DF, Vogl SE, Kaplan BH: Combination chemotherapy of advanced colorectal cancer with Triazinate and ICRF-159 after failure of 5-fluorouracil (Abstract). Proc Am Assoc Cancer Res 20:103, 1979
Vogl SE, Qazi R, Berenzwieg M: Combination chemotherapy for advanced colorectal cancer with triazinate, ICRF-159, 5-fluorouracil, and methyl-CCNU. Cancer Treat Rep 64:1143–1145, 1980
Marciniak TA, Moertel CG, Schutt AJ: Phase II study of ICRF-159 (NSC-129943) in advanced colorectal cancer. Cancer Chemother Rep 59:761–763, 1975
Gastrointestinal Tumor Study Group: Phase II trials of hexamethylmelamine, dianhydrogalactitol, razoxane and beta-2′-deoxythioguanosine as single agents against advanced measurable tumors of the pancreas. Cancer Treat Rep 69:713–716, 1985
Ahmann DL, Edmonson S, Frytak LK, Kuols LK, Bisel HF, Rubin J: Phase II study of ICRF-159 versus combination cis-DDP and DTIC in patients with disseminated malignant melanoma. Cancer Treat Rep 62:151–153, 1978
Dyment PG, Starling K, Land V: Razoxane (ICRF-159) in the treatment of pediatric solid tumors. Cancer Treat Rep 63:1397–1398, 1979
Ryall RD, Hanham WF, Newton A, Hellman K, Brinkley DM, Hjertaas OK: Combined treatment of soft tissue and osteosarcomas by radiation and ICRF-159. Cancer 34:1040–1045, 1974
Ryall RD, Bates T, Newton KA, Hellman K: Combination of radiotherapy and ICRF-159 for chondrosarcoma. Cancer 44:891–895, 1979
Corder MP, Tewfik HH, Clamon GH, Platz CE, Leimert JT, Herbst KD, Byfield JE: Radiotherapy plus Razoxane for advanced limited extent carcinoma of the lung. Cancer 53:1852–1856, 1984
Creighton AM, Hellman K, Whitecross S: Antitumor activity of a series of bisdiketopiperazines. Nature 222:384–395, 1969
Salsbury PJ, Burrage K, Hellman K: Inhibition of metastatic spread by ICRF-159: Selective depletion of a metastatic characteristic. Br Med J 4:344–346, 1970
Hellman K, Burrage K: Control of malignant metastases by ICRF-159. Nature 224:273–275, 1969
James SE, Salsbury AJ: Effect of (±)-1,2-Bis(3,5-dioxopiperazin-1-yl) propane on tumor blood vessels and its relationship to the antimetastatic effect in the Lewis lung carcinoma. Cancer Res 34:839–842, 1974
Atherton A: The effect of (±)1,2-bis(3,5-dioxopiperazin1-yl) propane (ICRF-159) on liver metastases from a hamster lymphoma. Eur J Cancer 11:383–388, 1975
Buin MV, Baldwin RW: Influence of ICRF-159 and Triton, W.R. 1339 on metastases of a rat epithelioma. Br J Cancer 13:62–67, 1975
Herman ELH, Mhatre RM, Chadwick DP: Modification of some of the toxic effects of daunomycin by pretreatment with ICRF-159. Toxicol Appl Pharmacol 27:517–526, 1974
Herman EH, Mhatre RM, Lee IP, Waravdekar VS: Prevenion of the cardiotoxic effects of adriamycin and daunomycin in the isolated dog heart (36432). Proc Soc Exp Biol Med 140:234–239, 1972
Woodman RJ, Cysyk RL, Kline I, Gang M, Venditti JM: Enhancement of the effectiveness of daunorubicin (NSC 82151) or adriamycin (NSC 123127) against early mouse L1210 leukemia with ICRF-159 (NSC 129943). Cancer Chemother Rep 59:689–695, 1975
Wampler GL, Speckhart V, Regelson W: Phase I clinical study of adriamycin ICRF-159 combination and other drug combinations (Abstract). Proc Am Assoc Cancer Res 15:189, 1974
Hellman K, Murkin GE: Synergism of ICRF-159 and radiotherapy in treatment of experimental tumors. Cancer 34:1033–1039, 1974
Poster DS, Penta JS, Bruno S, Macdonald JS: ICRF-187 in clinical oncology. Cancer Clin Trials 4:143–146, 1981
Herman E, Ardalan B, Bier C, Waravdekar V, Krop S: Reduction of daunomycin lethality and myocardial cellular alterations by pretreatment with ICRF-187 in Syrian Golden hamsters. Cancer Treat Rep 63:89–92, 1979
Levine BS, Henry MC, Port CD, Rosen E: Preclinical toxicologic evaluation of ICRF-187 in dogs. Cancer Treat Rep 64:1211–1215, 1980
Creighton AM, Birnie GD: Biochemical studies on growth-nhibitory bisdioxopiperazines. I. Effect on DNA, RNA and protein synthesis in mouse-embryo fibroblast. Int J Cancer 5:47–54, 1970
Sharpe HBA, Field FO, Hellman K: Mode of action of the cytostatic agent ‘ICRF-159’. Nature 226:524–526, 1970
Wheeler RH, Clauw DJ, Natale RB, Ruddon RW: The cytokinetic and cytotoxic effects of ICRF-159 and ICRF-187 in vitro and ICRF-187 in human bone marrow in vivo. Invest New Drugs 1:283–295, 1983
Hellman K, Field FO: Effect of ICRF-159 on the mammalian cell cycle: Significance for its use in cancer chemotherapy. J Natl Cancer Inst 44:539–543, 1970
LeServe AW, Hellman K: Metastases and the normalization of tumor blood vessels by ICRF-159: A new type of drug action. Br Med J 1:597–601, 1972
Koeller JM, Earhart RH, Davis HL: Phase I trial of ICRF-187 by 48 hours continuous infusion. Cancer Treat Rep 65:459–463, 1981
Liesmann J, Belt R, Haas C, Hoogstraten B: Phase I evaluation of ICRF-187 (NSC-169780) in patients with advanced malignancy. Cancer 47:1959–1962, 1981
VonHoff DD, Howser D, Lewis BJ, Holcenberg J, Weiss RB, Young RC: Phase I study of ICRF-187 using a daily for 3 days schedule. Cancer Treat Rep 65:249–252, 1981
Holcenberg JS, Tutsch KD, Earhart RH, Ungerleider RS, Kamen BS, Pratt CB, Gribble TJ, Glaubiger DL: Phase I study of ICRF-187 in pediatric cancer patients and comparison of its pharmacokinetics in children and adults. Cancer Treat Rep 70:703–709, 1986
Creaven PJ, Allen LM, Alford DA: The bioavailability in man of ICRF-159 — A new oral antineoplastic agent. J Pharm Pharmacol 27:914–918, 1975
Creaven PJ, Cohen MH, Hansen HH, Selawry OS, Taylor SG: Phase 1 clinical trial of a single dose and two weekly schedules of ICRF-159 (NSC-129943). Cancer Chemother Rep 58:393–400, 1974
Creaven PJ, Allen LM, Alford DA: The bioavailability in man of ICRF-159, a new oral antineoplastic agent. J Pharm Pharmacol 27:914–918, 1975
O'Connell MO, Begg CB, Silverstein MN, Glick JH, Oken MM: Randomized clinical trial comparing two dose regiments of ICRF-159 in refractory malignant lymphomas. Cancer Treat Rep 64:1355–1358, 1980
Sadee W, Starosoik J, Finn C, Cohen J: Determination of (+)-1,2-Bis(3–5-dioxopiperazinyl) propane plasma levels in rats, rabbits, and humans by GLC and mass fragmentography. J Pharm Sci 64:998–1001, 1975
Earhart RH, Tutsch KD, Koeller JM, Rodriguez R, Robins HI, Vogel CL, Davis HL, Tormey DC: Pharmacokinetics of (±)-1,2-Di(3,5-dioxopiperazin-1-yl) propane intravenous infusion in adult cancer patients. Cancer Res 42:5255–5261, 1982
Natale RB, Wheeler RH, Liepman MK, Sauder A, Bricker L: Phase II trial of ICRF-187 in non-small cell lung cancer. Cancer Treat Rep 67:311–313, 1983
Wheeler RH, Bricker LJ, Natale RB, Baker SR: A phase II trial of ICRF-187 in squamous cell carcinoma of the head and neck. Cancer Treat Rep 68:427–428, 1984
Arora M, Pazdur R, Samson M, Kinzie J, Baker L: ICRF-187 (NSC-168780): A phase II evaluation in metastatic colorectal carcinoma (Abstract). Proc Am Assoc Cancer Res 27:184, 1986
Brubaker LH, Vogel CL, Einhorn LH, Birch R: Treatment of advanced adenocarcinoma of the kidney with ICRF-187 (NSC 169780); a Southeastern Cancer Study group trial. Cancer Treat Rep 70:915–916, 1986
Joshi R, Smith B, Phillips RH, Barrett AJ: Acute myelomonocytic leukemia after Razoxane therapy. Lancet 2:1343–1344, 1981
Lakhani S, Davidson RN, Hiwaizi F, Marsden RA: Razoxane and leukemia. Lancet 2:288–289, 1984
Horton IJ, MacDonald DM, Wells RS: Epitheliomas in patients receiving Razoxane therapy for psoriasis. Br J Derm 109:675–678, 1983
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Vogel, C.L., Gorowski, E., Davila, E. et al. Phase I clinical trial and pharmacokinetics of weekly ICRF-187 (NSC 169780) infusion in patients with solid tumors. Invest New Drugs 5, 187–198 (1987). https://doi.org/10.1007/BF00203545
Issue Date:
DOI: https://doi.org/10.1007/BF00203545