Abstract
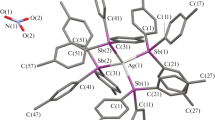
N,N′−Diphenylamidines and the silver(I) complexes of their deprotonated anions have been synthesized. Previously uncharacterized tetrameric structural motifs were produced by the inclusion of alkyl substituents at the amidinate carbon. The addition of a 2-methoxy functional group to the phenyl ring resulted in a cationic silver trimer in which hydrogen bonding links silver(I)-bound water molecules to the methoxy substituents. The thermal stabilities of the tetrameric species vary with alkyl chain length. The new complexes are: tetrakis(N,N′-diphenylpropamidinato) tetra silver(I), 1, tetrakis(N,N′-diphenylbutamidinato) tetrasilver(I) 2, tetrakis(N,N′-diphenylpentamidinato) tetra silver(I) 3, (N,N′-diphenyloctamidinato)silver(I) 4, (tetrakis(N,N′-di(4-n-butyl)phenylpropamidinato)tetrasilver(I), 5, bis(N,N′-di(2-methoxy)phenylacetamidinato)diaquatrisilver(I) nitrate 6 and tetrakis(N,N′-di(4-methoxy)phenylacetamidinato) tetrasilver(I), 7. Compounds 1, 5, 6 and 7 were structurally characterized by X-ray methods.
Similar content being viewed by others
REFERENCES
D. H. Klosterboer, Imaging Processes and Materials, Chapter 9, Neblette, 8th ed.
(a) The United Kingdom Chemical Database Service; D. A. Fletcher, R. F. McMeeking, and D. J. Parkin (1996). J. Chem. Info. Comput. Sci. 36, 746; (b) A. E. Blakeslee and J. L. Hoard (1956). J. Am. Chem. Soc. 78, 3029; (c) P. Coggon and A. T. McPhail (1972). J. Chem. Soc. Chem. Commun. 91; (d) R. G. Griffin, J. D. Ellett, Jr., M. Mehring, J. G. Bullitt, and J. S. Waugh (1972). J. Chem. Phys. 57, 2147; (e) V.-M. Hedrich and H. Hartl (1983). Acta. Cryst. C39, 533; (f ) T. C. W. Mak, W.-H. Yip, C. H. L. Kennard, G. Smith, and E. J. O'Reilly (1988). Aust. J. Chem. 41, 683; (g) G. Smith, D. S Sagatys, C. A. Campbell, D. E. Lynch, and C. H. L. Kennard (1990). Aust. J. Chem. 43, 1707; (h) X.-M. Chen, and T. C. W. Mak (1991). J. Chem. Soc., Dalton Trans. 1219; (i) P.-R. Wei, D.-D. Wu, and T. C. W. Mak (1996). Inorg. Chim. Acta 249, 169; ( j) B. P. Tolochko, S. V. Chernov, S. G. Nikitenko, and D. R. Whitcomb (1998). Nuclear Instruments and Methods in Physics Research, (A) 405, 428.
(a) J. Barker and M. Kilner (1994). Coord. Chem. Rev. 133, 219; (b) I. D. Brown and J. D. Dunitz (1961). Acta. Cryst. 14, 480; (c) J. Beck and J. Stra_ hle (1986). Z. Naturforsch. 41b, 4; (d) E. Hartmann and J. Strähle (1988). Z. Naturforsch. 43b, 818; (e) S. Maier, W. Hiller, J. Strähle, C. Ergezinger, and K. Dehnicke (1988). Z. Naturforsch. 43b, 1628; (f ) F. A. Cotton, X. Feng, M. Matusz, and R. Poli (1988). J. Am. Chem. Soc. 110, 7077; (g) D. Fenske, G. Baum, Zinn, and K. Dehnicke (1990). Z. Naturforsch. 45b, 1273.
(a) T. Ren, C. Lin, P. Amalberti, D. Mdacikenas, J. D. Protasiewicz, J. C. Baum, and T. L. Gibson (1998). Inorg. Chem. Commun. 1, 23; (b) S. J. Archibald, N. W. Alcock, and D. H. Busch, paper submitted.
E. Hartmann and J. Strähle (1989). Z. Naturforsch. 44b, 1.
J. Beck and J. Strähle (1986). Angew. Chem. Int. Ed. 25, 95.
J. E. O'Connor, G. A. Janusonis, and E. R. Corey (1968). J. Chem. Soc., Chem. Commun. 445.
E. Hartmann and J. Strähle (1990). Z. Anorg. Allg. Chem. 583, 31.
P. I. van Vliet, G. van Koten, and K. Vrieze (1979). J. Organomet. Chem. 179, 89.
S. Patai, The Chemistry of Amidines and Imidates (Wiley and Sons, 1975), Vol. 1.
E. C. Taylor and W. A. Erhart (1963). J. Org. Chem. 28, 1108; R. M. Roberts, R. H. De Wolfe, and J. H. Ross (1951). J. Am. Chem. Soc. 73, 2277; R. M. Roberts and R. H. De Wolfe, (1954). J. Am. Chem. Soc. 76, 2411; R. H. De Wolfe, Carboxylic Ortho Acid Derivatives (Academic Press, 1970).
F. B. Dains (1900). J. Am. Chem. Soc. 22, 181; F. B. Dains, R. C. Roberts, Brewster, and R. Q. (1916). J. Am. Chem. Soc. 38, 131.
S. P. Joshi, A. P. Khanolkar, and T. S. Wheeler (1936). J. Chem. Soc. 793; G. Mandel and A. J Hill (1954). J. Am. Chem. Soc. 76, 3978; A. J. Hill and J. V. Johnston (1954). J. Am. Chem. Soc. 76, 920.
SMART user's manual; Siemens Industrial Automation Inc. (Madison, WI, 1994); J. Cosier and A. M. Glazer (1986). J. Appl. Cryst. 19, 105-107.
G. M. Sheldrick (1990). Acta Cryst. A46, 467; G. M. Sheldrick, SHELX-96 (beta test), 1996.
For example: P. K. Mehrotra and R. Hofmann (1978). Inorg. Chem. 17, 2187; T. Yamaguchi and O. Lindqvist (1983). Acta Chem. Scand. A37, 685; B. Chiari, O. Piovesna, T. Tarantelli, and P. F. Zanazzi (1985). Inorg. Chem. 24, 366 and references therein; Y. Jiang, S. Alvarez, and R. Hofmann (1985). Inorg. Chem. 24, 749; K. M. Merz, Jr. and R. Hofmann (1988). Inorg. Chem. 27, 2120; A. Schäfer, C. Huber, G. J. Auss, and R. Ahlrichs (1993). Theor. Chim. Acta 87, 29; U. Siemeling, U. Vorfeld, B. Neumann, and H.-G. Stammler (1997). J. Chem. Soc., Chem. Commun. 1723; K. Singh, J. R. Long, and P. Stavropoulos (1997). J. Am. Chem. Soc. 119, 2942 and references therein; L. S. Ahmed, J. R. Dilworth, J. R. Miller, and N. Wheatley (1998). Inorg. Chim. Acta 278, 229; M. A. Omary, T. R. Webb, Z. Assefa, G. E. Shankie, and H. H. Patterson (1998). Inorg. Chem. 37, 1380; F. A. Cotton, X. Feng, and D. J. Timmons (1998). Inorg. Chem. 37, 4066; R. Villaneau, A. Proust, F. Robert, and P. G ouzerh (1998). J. Chem. Soc., Chem. Commun. 1491.
R. L. Griffith (1943), J. Chem. Phys. 11, 499.
A. Michaelides, V. Kiritsis, S. Skoulika, and A. Aubry (1993). Angew. Chem. Int. Ed. 32, 1495.
A. F. M. J. van der Ploeg, G. van Koten, and A. L. Spek (1979). Inorg. Chem. 18, 1052; L. M. Engelhardt, G. E. Jacobsen, W. C. Patalinghug, B. W. Skelton, C. L. Raston, and A. H. White (1991). J. Chem. Soc., Dalton Trans. 2859; J. A. Schuerman, F. R. Fronczek, and J. Selbin (1989). Inorg. Chim. Acta 160, 43.
For example: H. Schollhorn, U. Thewalt, and B. Lippert (1984). J. Chem. Soc., Chem. Commun. 769; X.-M. Chen and T. C. W. Mak (1991). J. Chem. Soc., Dalton Trans. 3253; N. J. Calos, C. H. L. Kennard, and T. C. W. Mak (1989). Aust. J. Chem. 42, 2047.
C.-M. Che, H.-K. Yip, V. W.-W. Yam, P.-Y. Cheung, T.-F. Lai, S.-J. Shieh, and S.-M. Peng (1992). J. Chem. Soc., Dalton Trans. 427.
H. H. Murray, R. G. Raptis, and J. P. Fackler (1988). Inorg. Chem. 27, 26.
A. J. Amoroso, J. C. Jeffery, P. L. Jones, J. A. McCleverty, E. Psilakis, and M. D. Ward (1995). J. Chem. Soc., Chem. Commun. 1175; E. R. Humphrey, N. C. Harden, L. H. Rees, J. C. Jeffery, J. A. McCleverty, and M. D. Ward (1998). J. Chem. Soc., Dalton Trans. 3353.
C.-M. Che, H.-K. Yip, D. Li, S.-M. Peng, G.-H. Lee, Y.-W. Wang, and S.-T. Liu (1991). J. Chem. Soc., Chem. Commun. 1615.
C. King, J. C. Wang, N. I. M. Khan, and J. P. Fackler (1989). Inorg. Chem. 28, 2145.
M. Kilner and A. Pietrzykowski (1983). Polyhedron 2, 1379.
E. K. Fields and S. Meyerson (1976). J. Org. Chem. 41, 916.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Archibald, S.J., Alcock, N.W., Busch, D.H. et al. Synthesis and Characterization of Silver(I) Complexes with C-Alkyl Functionalized N,N′-Diphenylamidinates: Tetrameric and Trimeric Structural Motifs. Journal of Cluster Science 11, 261–283 (2000). https://doi.org/10.1023/A:1009085319864
Issue Date:
DOI: https://doi.org/10.1023/A:1009085319864