Abstract
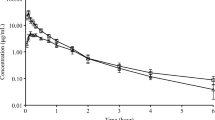
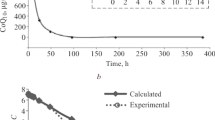
The pharmacokinetics and pharmacodynamics of L-703,014, a fibrinogen receptor antagonist, have been examined in the dog. An analytical method which utilizes methanol precipitation of dog plasma proteins followed by HPLC with an automated column switching technique using the chemical analogue L-704,326 as internal standard was developed for the determination of L-703,014 in dog plasma. The compound was not metabolized in the dog and was eliminated in the kidneys and into bile. Of the administered dose, 68.9 ± 1.3% (i.v.) and 80.5 ± 11.9% (p.o.) were recovered in the feces; 20.3 ± 3% (i.v.) and 2.2 ± 0.2% (p.o.) were recovered in the urine by 72 hr. L-703,014 was 23 ± 3.4% bound in dog plasma protein and the mean ratio of plasma/whole blood was 1.22 ± 0.05. The mean terminal half-life was 118 ± 36 min, the mean steady-state volume of distribution was 0.61 ± 0.22 L/kg, and the mean plasma clearance was 8 ± 2 mL/min/kg. Ex vivo platelet aggregation measurements were made by inducing platelet aggregation with 10 µg/ mL collagen in the presence of 1 µM epinephrine as an agonist. The mean C 50 was 44.4 ± 6.0 ng/mL, and the mean Hill coefficient was 1.5 ± 0.3. The mean bioavailability was 4.9 ± 1.4% in dogs administered 2.0 mg/kg (p.o.).
Similar content being viewed by others
REFERENCES
E. R. Bates, M. J. McGillem, J. K. Mickelson, B. Pitt, and G. B. J. Mancini. A monoclonal antibody against platelet glycoprotein IIb/IIIa receptor complex prevents platelet aggregation and thrombosis in a canine model of coronary angioplasty. Circulation 84:2463–2469 (1991).
P. S. Wissel, J. S. Barrett, T. D. Bjornsson, A. Barchowsky, R. J. Gould, E. Hand, D. Farrell, and D. Panebianco. Pharmacokinetics and pharmacodynamic effects of MK-852, a platelet fibrinogen receptor antagonist. Pharm. Res. 9:S293 (1992).
A. K. Pedersen and G. A. Fitzgerald. The human pharmacology of platelet inhibition: Pharmacokinetics relevant to drug action. Circulation 72:1164–1176 (1985).
J. Mordenti and A. Rescigno. Theoretical considerations for the design and development of a drug: Influence of pharmacokinetics and dosage regimen on receptor interactions. Pharm. Res. 10:777–782 (1993).
G. D. Hartman, M. S. Egbertson, W. Halezenko, W. L. Laswell, M. E. Duggan, R. L. Smith, A. M. Naylor, P. D. Manno, R. J. Lynch, G. Zhang, C. Chang, and R. J. Gould. Non-peptide fibrinogen receptor antagonists. 1. Discovery and design of exosite inhibitors. J. Med. Chem. 35:4640–4642 (1992).
Z.-R. Gan, R. J. Gould, J. W. Jacobs, P. A. Friedman, and M. A. Polokoff. Echistatin. A potent platelet aggregation inhibitor from the venom of the viper, Echis carinatus. J. Biol. Chem. 263:19827–19832 (1988).
SAS Institute, Inc. SAS/STAT User's Guide, Vol. 2, Version 6, 4th Ed. SAS Institute, Cary, NC, 1990.
D. Perrier and M. Mayersohn. Noncompartmental determination of the steady-state volume of distribution for any mode of administration. J. Pharm. Sci. 71:372–373 (1982).
C. Picard-Fraire. Ticlopidine hydrochloride: Relation between dose, kinetics, plasma concentration and effect on platelet function. Thromb. Res. (Suppl.) 4:119–128 (1983).
C. Picard-Fraire. Pharmacokinetic and metabolic characteristics of ticlopidine in relation to its inhibitory properties on platelet function. Agents Actions 15(Suppl):68–75 (1984).
J. J. Thebault, C. E. Blatrix, J. F. Blanchard, and E. A. Panak. Effects of ticlopidine, a new platelet aggregation inhibitor in man. Clin. Pharmacol. Ther. 18:485–490 (1975).
T. DiPerri, F. L. Pasini, C. Frigerio, P. Blardi, F. Centini, G. L. Messa, A. Ghezzi, and L. Volpi. Pharmacodynamics of ticlopidine in man in relation to plasma and cell concentration. Eur. J. Clin. Pharmacol. 41:429–434 (1991).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Barrett, J.S., Gould, R.J., Ellis, J.D. et al. Pharmacokinetics and Pharmacodynamics of L-703,014, a Potent Fibrinogen Receptor Antagonist, After Intravenous and Oral Administration in the Dog. Pharm Res 11, 426–431 (1994). https://doi.org/10.1023/A:1018973323039
Issue Date:
DOI: https://doi.org/10.1023/A:1018973323039