Abstract
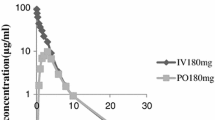
Cefixime (CL 284,635; FK 027) is a new third-generation oral cephalosporin. To study dose-dependent pharmacokinetics of cefixime in dogs, two balanced four-way crossover studies were conducted. In the first study, oral doses of 50, 100, and 200 mg/kg and an intravenous dose of 50 mg/kg cefixime were administered. In the second study, oral doses of 6.25, 12.5, and 25 mg/kg and an intravenous dose of 12.5 mg/kg cefixime were administered to the same dogs. A period of 1 month separated the two studies. When the two intravenous doses were compared (i.e., 12.5 and 50 mg/kg), a twofold increase in clearance and volume of distribution was observed after the higher dose. The oral systemic bioavailability in the dose range 6.25–50 mg/kg was 55%. It decreased to 44% at 100 mg/kg and 27% at 200 mg/kg. The average peak serum concentrations ranged from 15.8 µg/ml at 6.25 mg/kg to 119 µg/ml at 200 mg/kg. Within this concentration range, the fraction of free drug in serum (unbound to proteins) increased from 7 to 25%. This concentration-dependent protein binding was primarily responsible for changes in total clearance, volume of distribution, and bioavailability of the drug in dogs.
Similar content being viewed by others
REFERENCES
Y. Shigi, Y. Matsumoto, M. Kaizu, Y. Fijishita, and H. H. Kajo. J. Antibiot. 37:790–796 (1984).
T. Kanimura, H. Kojo, Y. Matsumoto, Y. Mine, S. Goto, and S. Kuwahara. Antimicrob. Agents Chemother. 25:98–104 (1984).
H. C. Neu and P. Labthavikul, 23rd Intersci. Conf. Antimicrob. Agents Chemother., Abstr. 256 (1983).
T. Hirose, H. Sakamoto, and Y. Mine. 23rd Intersci. Conf. Antimicrob. Agents Chemother., Abstr. 256C (1983).
A. P. Tonelli, M. Bialer, and A. Yacobi. J. Pharm. Sci. 74:1242–1244 (1985).
H. Sakamoto, T. Hirose, and Y. Mine. J. Antibiot. 38:496–504 (1985).
V. K. Batra, M. Bialer, J. A. Morrison, B. M. Silber, and A. Yacobi, Abstracts of the 132nd Meeting, American Pharmaceutical Association, San Antonio, p. 116 (1985).
M. Nakashima and K. Ueno. 23rd Intersci. Conf. Antimicrob. Agents Chemother., Abstr. 265A (1983).
M. Bialer, A. P. Tonelli, J. D. Kantrowitz, and A. Yacobi. Drug Metab. Dispos. 14:132–136 (1986).
A. J. Falkowski, Z. M. Look, R. J. Kelly, B. M. Silber, and A. Yacobi. Isocratic elution reverse phase liquid chromatographic determination of a new cephalosporin compound in biological samples (submitted for publication).
A. J. Falkowski, Z. M. Look, R. J. Kelly, B. M. Silber, and H. Noguchi, Abstracts of the 37th National Meeting, Academy of Pharmaceutical Sciences, Philadelphia, p. 238 (1984).
Y. Tokuma, Y. Shiozaki, and H. Noguchi. J. Chromatogr. Biomed. Appl. 311:339–346 (1984).
M. Gibaldi and D. Perrier. Pharmacokinetics, 2nd ed., Marcel Dekker, New York, 1982, pp. 1–43, 445–449.
D. S. Riggs. The Mathematical Approach to Physiological Problems, William and Wilkins, Baltimore, 1983, pp. 168–220.
M. Gibaldi and D. Perrier. Pharmacokinetics, 2nd ed., Marcell Dekker, New York, 1982, pp. 409–417.
P. L. Altman and D. S. Dittmer. Biology Data Book, Vol. II, 2nd ed., Fed. Am. Soc. Exp. Biol., Bethesda, Md., 1974, pp. 1701–1706.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Bialer, M., Batra, V.K., Morrison, J.A. et al. Dose-Dependent Pharmacokinetics of a New Oral Cephalosporin, Cefixime, in the Dog. Pharm Res 4, 33–37 (1987). https://doi.org/10.1023/A:1016473709720
Issue Date:
DOI: https://doi.org/10.1023/A:1016473709720