Summary
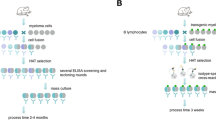
Non-obese diabetic mice display a syndrome with dramatic clinical and pathological features similar to those of Type 1 (insulin-dependent) diabetes in man. Circulating autoantibodies to the surface of islet cells were demonstrated in some of these mice by a protein A radioligand assay. To produce monoclonal antibodies to islet cell surface antigens, therefore, we took the spleens of non-obese diabetic mice, transferred the spleen cells into non-immunized recipient mice, which were made immunologically incompetent by a large dose of X-irradiation, and then fused their lymphocytes with FO mouse myeloma cells. After screening the resultant hybrids, one stable hybridoma (3A4) that produced a monoclonal antibody (IgG1) specifically bound to the surface of islet cells was obtained. The purified monoclonal antibody was bound to the surface of transplantable Syrian golden hamster insulinoma cells sevenfold more than control antibody. Adsorption of the antibody on mouse spleen lymphocytes or thymocytes resulted in only a slight decrease in 125I-protein A binding to insulinoma cells. This antibody also reacted with the surface of mouse and rat islet cells, but not with that of rat spleen cells or hepatocytes. A spectrophotometric assay for peroxidase activity demonstrated that six times more peroxidase bound to insulinoma cells incubated with the antibody than to cells treated with control antibody. Furthermore, this antibody could be visually detected in the immunoenzymatic labelling of the surface of insulinoma cells. In summary, we have developed a novel method of producing monoclonal antibodies to the surface of islet cells for probing into the pathogenesis of Type 1 diabetes.
Article PDF
Similar content being viewed by others
References
Gepts W (1965) Pathologic anatomy of the pancreas in juvenile onset diabetes. Diabetes 14: 619–639
Gepts W, Le Compte PM (1981) The pancreatic islets in diabetes. Am J Med 70:105–115
Bottazzo GF, Florin-Christensen A, Doniach D (1974) Islet cell antibodies in diabetes mellitus with autoimmune polyendocrine deficiencies. Lancet 2: 1279–1283
MacCuish AC, Jordan J, Campbell CJ, Duncan LJP, Irvine WJ (1974) Antibodies to islet-cells in insulin-dependent diabetics with coexistent autoimmune disease. Lancet 2:1529–1533
Lernmark Å, Freedman ZP, Hoffmann C, Rubenstein AH, Steiner DF, Jackson RJ, Traisman HS (1978) Islet cell surface antibodies in juvenile diabetes mellitus. N Engl J Med 299: 375–380
Eisenbarth GS, Oie H, Gazdar A, Chick W, Schultz JA, Scearce RM (1981) Production of monoclonal antibodies reacting with rat islet cell membrane antigens. Diabetes 30: 226–230
Makino S, Kunimoto K, Muraoka Y, Mizushima Y, Katagiri K, Tochino Y (1980) Breeding of a non-obese diabetic strain of mice. Exp Anim 29: 1–13
Makino S, Kunimoto K, Muraoka Y, Katagiri K (1980) Effect of castration on the appearance of diabetes in the NOD mouse. Exp Anim 30: 137–140
Nakhooda AF, Like AA, Chappel CL, Murray FT, Marliss EB (1977) The spontaneously diabetic Wistar rat. Metabolic and morphologic studies. Diabetes 26:100–112
Fazekas de St. Groth S, Scheidegger D (1980) Production of monoclonal antibodies. Strategy and tactics. J Immunol Methods 35: 1–21
Uchida S, Watanabe S, Aizawa T, Furuno A, Muto T (1979) Polyoncogenicity and insulinoma inducing ability of BK virus, a human papovavirus, in Syrian golden hamsters. J Natl Cancer Inst 63: 119–126
Lernmark Å (1974) The preparation of and studies on free cell suspensions from mouse pancreatic islets. Diabetologia 10: 431–438
Dobersen M, Scharff J, Notkins A (1980) Microculture system for studying monolayers of functional B-cells. Endocrinology 106: 1070–1073
Matsumura T, Nitta K, Yoshikawa M, Takaoka T, Katsuta H (1975) Action of bacterial neutral protease on the dispersion of mammalian cells in tissue culture. Jpn J Exp Med 45: 383–392
Seglen PO (1973) Preparation of rat liver cells. III. Enzymatic requirements for tissue dispersion. Exptl Cell Res 82: 391–398
Hamaoka T, Kitagawa M, Matsuoka Y, Yamamura Y (1969) Antibody production in mice. I. The analysis of immunological memory. Immunology 17: 55–69
Oi VT, Herzenberg LA (1980) Immunoglobulin-producing hybrid cell lines. In: Mishell BB, Shiigi SM (eds) Selected methods in cellular immunology, Vol 17. Freeman, San Francisco, pp 351–372
Lowry HO, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 53: 265–275
Lernmark Å, Kanatsuna T, Patzelt C, Diakoumis K, Carroll R, Rubenstein AH, Steiner DF (1980) Antibodies directed against the pancreatic islet cell plasma membrane. Detection and specificity. Diabetologia 19: 445–451
Roth RA, Rotman B (1975) Inactivation of normal β-D-galactosidase by antibodies to defective forms of the enzyme. J Biol Chem 250: 7759–7765
Jonak ZL (1980) Peroxidase-conjugated antiglobulin method for visual detection of cell-surface antigens. In: Kennet RH, McKearn TJ, Bechtol KB (eds) Monoclonal antibodies. Plenum Press, New York, pp 378–380
Köhler G, Shulman MJ (1978) Cellular and molecular restrictions of the lymphocyte fusion. Curr Top Microbiol Immunol 81: 143–148
Mosmann TR, Longenecker BM (1982) The high background immune reactivity of mice to polymorphic determinants on xenogenetic erythrocytes: theoretical and practical implications. J Immunol 128: 100–104
Till JE, McCulloch EA (1961) A direct measurement of the radiation sensitivity of normal mouse bone marrow cells. Radiat Res 14: 213–222
Fraser CM, Venter JC (1980) Monoclonal antibodies to β-adrenergic receptors: Use in purification and molecular characterization of β receptors. Proc Natl Acad Sci USA 77: 7034–7038
Myles DG, Primakoff P, Bellvé AR (1981) Surface domains of the guinea pig serum defined with monoclonal antibodies. Cell 23: 433–439
Stähli C, Staehelin T, Miggiano V, Schmidt J, Häring P (1980) High frequencies of antigen-specific hybridomas. Dependence on immunization parameters and prediction by spleen cell analysis. J Immunol Methods 32: 297–304
Negishi K (1983) Quantitative estimation of islet cell surface antibodies using 125I-antihuman IgG antibody and insulinoma cells as target cells, and its clinical application. J Japan Diab Soc 26: 485–496
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Yokono, K., Shii, K., Hari, J. et al. Production of monoclonal antibodies to islet cell surface antigens using hybridization of spleen lymphocytes from non-obese diabetic mice. Diabetologia 26, 379–385 (1984). https://doi.org/10.1007/BF00266041
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00266041