Abstract
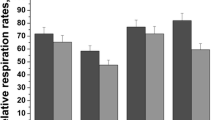
1) Aldehyde dehydrogenase with a Km for acetaldehyde in the micromolar range (referred to as low Km-AldDH) is located in the matrix and that with a Km in the millimolar range (referred to as high Km-AldDH) is located in both the matrix and the outer membrane of mitochondria. 2) Pargyline in vivo treatment (100 mg/kg i.p.) caused a complete inhibition of the low Km-AldDH activity of the matrix fraction but caused only a 10% inhibition of the high Km-AldDH activities of the matrix and the outer membrane fractions. Diethyldithiocarbamate (DDC) treatment (700 mg/kg i.p.) caused less than 80% inhibition of the low Km-AldDH activity of the matrix fraction and caused a 20% inhibition of the high Km-AldDH activity of the matrix fraction. DDC treatment had no effect on the enzyme activity of the outer membrane fraction. The difference in the degree of inhibition of the low Km-AldDH activity of the matrix fraction by pargyline and DDC treatment may cause different blood acetaldehyde levels during ethanol oxidation.
Similar content being viewed by others
References
Cohen G, MacNamee D, Dembiec D (1975) Elevation in blood acetaldehyde by pargyline during ethanol administration. Biochem Pharmacol 24: 313–316
Dembiec D, MacNamee D, Cohen G (1976) The effect of pargyline and other monoamine oxidase inhibitors on blood acetaldehyde levels in ethanol-intoxicated mice. J Pharmacol Exp Ther 197: 332–339
Greenawalt JW (1974) The isolation of outer and inner mitochondrial membranes. In: Fleischer S, Packer L (eds) Methods in enzymology, vol 31. Academic Press, New York San Francisco London, pp 310–323
Lebsak ME, Petersen DR, Collins AC, Anderson AD (1977) Preferential inhibition of the low Km aldehyde dehydrogenase activity by pargyline. Biochem Pharmacol 26: 1151–1154
Lineweaver H, Burk D (1934) The determination of enzyme dissociation constants. J Am Chem Soc 56: 658–666
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193: 265–275
Nakanishi S, Shiohara E, Tsukada M, Iijima Y (1977) Aldehyde dehydrogenase activities after treatment with phenobarbital in Long-Evans rats. Jpn J Stud Alcohol 12: 5–12
Parrilla R, Ohkawa K, Lindros KO, Zimmerman U-JP, Kobayashi K, Williamson JR (1974) Functional compartmentation of acetaldehyde oxidation in rat liver. J Biol Chem 249: 4926–4933
Reiner JM (1969) The analysis of complex biological systems. In: Behavior of enzyme systems, 2nd ed. Van Nostrand-Reinhold Co, New York Toronto London Melbourne, pp 108–132
Schnaitman C, Greenawalt JW (1968) Enzymatic properties of the inner and outer membranes of rat liver mitochondria. J Cell Biol 38: 158–175
Schnaitman C, Erwin VG, Greenawalt JW (1967) The submitochondrial localization of monoamine oxidase. An enzymatic marker for the outer membrane of rat liver mitochondria. J Cell Biol 32: 719–735
Smith L (1974) Spectrophotometric assay of cytochrome c oxidase. In: Bergmeyer HU (ed) Methods of biochemical analysis, 2nd ed, vol II. Verlag Chemie, Weinheim pp 427–434
Tottmar SOC, Pettersson H, Kiessling K-H (1973) The subcellular distribution and properties of aldehyde dehydrogenases in rat liver. Biochem J 135: 577–586
Tottmar SOC, Pettersson H, Kiessling K-H (1974) Aldehyde dehydrogenases in rat liver. In: Thurman RG, Yonetani T, Williamson JR, Chance B (eds) Alcohol and aldehyde metabolizing system. Academic Press, New York London, pp 147–160
Yamazaki H, Nishiguchi K, Nakanishi S (1979) Effects of some drugs on aldehyde dehydrogenase activities of submitochondrial fractions of rat liver. Jpn J Stud Alcohol 14: 328
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Nakanishi, S., Yamazaki, H., Nishiguchi, K. et al. Effects of pargyline and diethyldithiocarbamate in vivo treatment on aldehyde dehydrogenase activities of submitochondrial fractions. Arch Toxicol 46, 241–248 (1980). https://doi.org/10.1007/BF00310440
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00310440