Summary
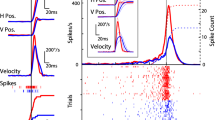
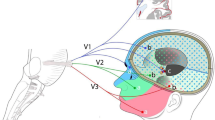
The tectofugal pathway in birds has been reported to process primarily information from the contralateral eye. Although this pathway has access to the contralateral hemisphere by various connections, electrophysiological recordings up to now have failed to demonstrate any excitatory influence of visual stimulation in the higher stations of this pathway. This study is the first to demonstrate an excitatory projection from the ipsilateral eye to the telencephalic projection area of the tectofugal pathway by recordings of visually evoked potentials in the ectostriatum. The excitatory projection probably leads from the eye to the contralateral tectum opticum, then recrosses back to the nucleus rotundus of the ipsilateral side where it reaches the ectostriatum. In normal birds, the ipsilateral stimulus responses in the ectostriatum are smaller in amplitude and have a longer latency than responses to contralateral stimuli. In unilaterally enucleated birds, the ipsilateral response is enhanced in the ectostriatum and can be detected in the nucleus rotundus, too. The results suggest that in normal birds the ipsilateral response is inhibited to a high degree by spontaneous activity of the contralateral eye. Possibly, this counterbalanced inhibition provides a mechanism for weighting information from the left and right eye field in order to ensure adequate processing of stimuli.
Similar content being viewed by others
References
Bagnoli P, Grassi S, Magni F (1977) Visual wulst influences on the optic tectum of the pigeon. Brain Behav Evol 14: 217–237
Bagnoli P, Francesconi W, Magni F (1979) Interaction of optic tract and visual wulst impulses on single units of the pigeon's optic tectum. Brain Behav Evol 16: 19–37
Bagnoli P, Grassi S, Magni F (1980) A direct connection between visual wulst and tectum opticum in the pigeon (Columba Livia) demonstrated by horseradish peroxidase. Arch Ital Biol 118: 72–88
Bagnoli P, Burkhalter A (1983) Organisation of the afferent projections to the wulst in the pigeon. J Comp Neurol 214: 103–113
Benowitz LJ, Karten HJ (1976) Organisation of the tectofugal visual pathway in the pigeon. A retrograde transport study. J Comp Neurol 167: 503–520
Bischof HJ (1981) A stereotaxic headholder for small birds. Brain Res Bull 7: 435–436
Bons N (1976) Retinohypothalamic pathway in the duck (Anas platyrhynchos). Cell Tiss Res 168: 343–360
Cohen DH, Karten HJ (1974) The structural organisation of the avian brain: an overview. In: Goodman IJ, Schein MW (eds) Birds, brain and behaviour. Academic Press, New York, pp 29–73
de Britto LRG, Brunelli M, Francesconi W, Magni F (1975) Visual response pattern of thalamic neurons in the pigeon. Brain Res 97: 337–343
Denton CJ (1981) Topography of the hyperstriatal visual projection area in the young domestic chicken. Exp Neurol 74: 482–498
Ehrlich D, Mills D (1985) The effects of binocular and monocular occlusion on the number of optic nerve axons containing degenerative organelles. Neurosci Lett 54: 351–356
Emmerton J (1983) Functional morphology of the visual system. In: Abs M (ed) Physiology and behaviour of the pigeon. Academic Press, London, pp 221–244
Hardy O, Leresche N, Jassik-Gerschenfeld D (1984) Postsynaptic potentials in neurons of the pigeon's opticum tectum in response to afferent stimulation from the retina and other visual structures: an intracellular study. Brain Res 311: 65–74
Holden AL (1968a) The field potential profile during activation of the avian optic tectum. J Physiol (Lond) 194: 75–90
Holden AL (1968b) Types of unitary response and correlation with the field potential profile during activation of the avian optic tectum. J Physiol (Lond) 194; 91–106
Hunt SP, Künzle H (1976) Observations of the projections and intrinsic organisation of the pigeon optic tectum: an autoradiographic study based on anterograde and retrograde axonal and dendritic flow. J Comp Neurol 170: 153–172
Karten HJ (1969) The organisation of the avian telencephalon and some speculations on the phylogeny of the amniote telencephalon. Ann NY Acad Sci 167: 164–179
Karten HJ, Hodos W, Nauta WJH, Revzin AM (1973) Neural connections of the ‘visual wulst’ of the avian telencephalon. Experimental studies in the pigeon (Columba livia) and owl (Speotyto cunicularia). J Comp Neurol 150: 253–278
Llinás R, Nicholson C (1974) Analysis of field potentials in the central nervous system. In: Stevens CF (ed) Handbook of EEG and clinical neurophysiology. Elsevier, Amsterdam, pp 62–92
Leresche N, Hardy O, Audinat E, Jassik-Gerschenfeld D (1986) Synaptic transmission of excitation from the retina to cells in the pigeon's optic tectum. Brain Res 365: 138–144
McLoon SC (1982) Alterations in precision of the crossed retinotectal projection during chick development. Science 215: 1418–1420
McLoon SC, Lund RD (1982) Transient retinofugal pathways in the developing chick. Exp Brain Res 45: 277–284
Mihailovic J, Perisic M, Bergonzi R, Meier RE (1974) The dorsolateral thalamus as a relay in the retino-wulst pathway in pigeon (Columba livia). Exp Brain Res 21: 229–240
Mori S (1973) Analysis of field response in optic tectum of the pigeon. Brain Res 54: 193–206
Nixdorf BN, Bischof HJ (1982) Afferent connections of the ectostriatum and visual wulst in, the zebra finch (Taeniopygia guttata castanotis Gould) a HRP study. Brain Res 248: 9–17
O'Leary DDM, Gerfen CR, Cowan WM (1983) The development and restriction of the ipsilateral retinofugal projection in the chick. Dev Brain Res 10: 93–109
Parker DM, Delius JD (1972) Visual evoked potentials in the forebrain of the pigeon. Exp Brain Res 14: 198–209
Perisic M, Mihailovic J, Cuénod M (1971) Electrophysiology of contralateral and ipsilateral visual projections to the wulst in pigeon (Columba livia). Int J Neurosci 2: 7–14
Pettigrew JD, Konishi M (1976) Neurons selective for orientation and binocular disparity in the visual wulst of the barn owl (Tyto alba). Science 193: 675–678
Revzin AM (1969) A specific visual projection area in the hyperstriatum of the pigeon. Brain Res 15: 246–249
Revzin AM (1970) Some characeristics of wide field units in the brain of the pigeon. Brain Behav Evol 3: 195–204
Revzin AM, Karten HJ (1966/1967) Rostral projections of the optic tectum and the nucleus rotundus in the pigeon. Brain Res 3: 264–276
Ritchie TC, Cohen DH (1977) The avian tectofugal visual pathway: projections of its telencephalic target, the ectostriatal complex. Soc Neurosci Abstr 3: 94
Robert F, Cuénod M (1969a) Electrophysiology of the intertectal commissures in the pigeon. I. Analysis of the pathways. Exp Brain Res 9: 116–121
Robert F, Cuénod M (1969b) Electrophysiology of the intertectal commissures in the pigeon. II. Inhibitory interaction. Exp Brain Res 9: 123–136
Takatsuji K, Hironobu I, Masai H (1983) Ipsilateral retinal projections in Japanese quail, Coturnix coturnix japonica. Brain Res Bull 10: 53–56
Weidner C, Reperant J, Miceli D, Haby M, Rio JR (1985) An anatomical study of ipsilateral retinal projection in the quail using radioautographic, horseradish peroxidase, fluorescence and degeneration techniques. Brain Res 340: 99–108
Watanabe M, Ito H, Ikushima M (1985) Cytoarchitecture and ultrastructure of the avian ectostriatum: afferent terminals from the dorsal telencephalon and some nuclei in the thalamus. J Comp Neurol 236: 241–257
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Engelage, J., Bischof, H.J. Enucleation enhances ipsilateral flash evoked responses in the ectostriatum of the zebra finch (Taeniopygia guttata castanotis Gould). Exp Brain Res 70, 79–89 (1988). https://doi.org/10.1007/BF00271850
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00271850