Summary
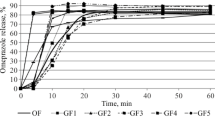
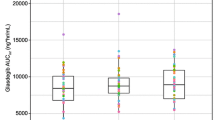
Three different pivmecillinam preparations, a conventional 200 mg tablet (P tablet) and two new formulations containing respectively pivmecillinam 200 mg and 400 mg plus Avicel® (microcrystalline cellulose) as a disintegrator (PA tablet), were compared in vitro and in a gastroscopic study in 8 healthy volunteers. Disintegration of the PA tablet was significantly more rapid both in vitro and in the stomach. Following disintegration, the content of the PA tablet was spread over a larger area of the gastric mucosa (1088 mm2) than was observed with the P tablets (408 mm2). Three of the 8 volunteers taking the P tablet developed hyperaemia, interstitial bleeding or erosions of the mucosa of the stomach. No such reactions were seen with the PA tablets. Serum concentrations of mecillinam following ingestion of pivmecillinam tablets were determined in three groups of subjects; fasting volunteers, both supine and ambulant, and in ambulant subjects who took the preparation with a light meal. There was a tendency for the new PA tablets to produce a higher peak serum level as well as greater bioavailability of mecillinam. Administration of the PA tablets with a meal significantly increased the peak serum level and total bioavailability of the drug. On the basis of our observations we recommend adoption of the new PA tablet, because of its quick passage through the oesophagus and its more rapid and complete disintegration in the stomach.
Similar content being viewed by others
References
Abbot FK, Mack M, Wolf SG (1952) The relation of sustained contraction of the duodenum to nausea and vomiting. Gastroenterology 20: 238
Bokey L, Hugh T (1975) Oesophagical ulceration associated with doxycycline therapy. Med J Aust 1: 236–237
Carlborg B, Kumlien A, Olsson H (1978) Medikamentella esofagusstrikurer. Läkartidningen 75: 4609–4611
Cronberg S, Egberg M, Hjorth N, Lindblom EP, Nilsson T, Nordmark B, Parment PA, Renmarker K, Richtoff SJ, Skog P (1980) Pivmecillinambehandling vid akut cystit — randomiserad jämförelse mellan doseringar. Läkartidningen 77: 2701–2702
Cronstedt J, Carling L, Vestergaard P, Berglund J (1978) Oesophageal disease revealed by endoscopy in 1000 patients referred primarily for gastroscopy. Acta Med Scand 204: 413–416
Crowson TD, Head LH, Ferrante WA (1976) Esophageal ulcers associated with tetracycline therapy. J Am Med Assoc 235: 2747–2748
Engberg-Pedersen H (1974) Empirical equation for pharmacokinetics analysis of drug serum levels. Antimicrob Agents Chemother 6: 554–562
Flodström A, Hemlin C (1980) Ännu ett läkemedel med risk for esofagusskador? Läkartidningen 77: 501
Griffith GH, Owea GM, Campelt PH, Shields R (1968) Gastric emptying in health and gastroduodenal disease. Gastroenterology 54: 1–7
Hansen PH, Kristensen KH, Lenler-Eriksen HA, Pagh J, Østergård JE (1981) Pivmecillinam (Selexid®) in the treatment of acute cystitis. Comparison between three and seven days treatment Ugeskr Laeg 143: 670–673
Hey H, Matzen P, Thorup Andersen J (1979a) A gastroscopic and pharmacological study of the disintegration time and absorption of pivampicillin capsules and tablets. Br J Clin Pharmacol 8: 237–242
Hey H, Matzen P, Andersen JT, Didriksen E, Nielsen B (1979b) The absorption and tolerability of various pivampicillin formulations. Arch Pharm Chem 7: 169–174
Hollander M, Wolfe DA (1973) Nonparametric statistical methods. J Wiley and Sons, New York, London
Pemberton J (1970) Oesophageal obstruction and ulceration caused by oral potassium therapy. Br Heart J 32: 267–268
Roholt K, Nielsen B, Kristensen E (1975) Pharmacokinetic studies with mecillinam and pivmecillinam. Chemotherapy 21: 146–166
Thorup Andersen J, Secher-Hansen E (1982) A method for determination of linear sizes and areas during endoscopy. Dan Med Bull in press
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Hey, H., Frederiksen, H.J. & Thorup Andersen, J. Gastroscopic and pharmacokinetic evaluation of a new pivmecillinam tablet. Eur J Clin Pharmacol 22, 63–69 (1982). https://doi.org/10.1007/BF00606427
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00606427