Summary
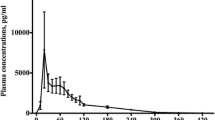
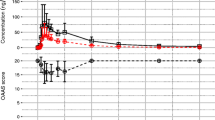
The oral bioavailability of high-dose metoclopramide was studied in 12 patients, who received oral or intravenous (i.v.) metoclopramide in random order with each of 2 consecutive courses of cytotoxic chemotherapy. The terminal half-life of metoclopramide was 7.1±0.4 h (mean±SEM) and was not affected by the route of drug administration. Mean bioavailability was 86.6±4.7% and the range (65–118%) was less than that reported for standard doses. Neither half-life nor bioavailability was significantly correlated with age. Adverse effects were mild and were similar for both oral and iv metoclopramide. Oral high-dose metoclopramide, given in the same doses as for i.v. administration, should therefore be as effective as the i.v. regimen and may be easier to administer.
Similar content being viewed by others
References
Alavi JB, Torri S, Glick JH (1984) High dose oral metoclopramide: an effective antiemetic agent. Proc Am Soc Clin Oncol 3: 109
Anthony LB, Krozely MG, Woodward NJ, Hainsworth JD, Hande KR, Brenner DE, Greco FA, Burish TG (1984) Antiemetic effect of oral vs intravenous metoclopramide in patients receiving cisplatinum: A randomized double-blind trial. Proc Am Soc Clin Oncol 3: 104
Bateman DN, Kahn C, Davies DS (1980) The pharmacokinetics of metoclopramide in man with observations in the dog. Br J Clin Pharmacol 9: 371–377
Block W, Pingoud A, Khan M, Kjellerup P (1981) The pharmacokinetics, bioequivalence and bioavailability of different formulations of metoclopramide in man. Arzneimittelforsch Drug Res 31: 1041–1045
Garnick MB (1983) Oral metoclopramide and cisplatin chemotherapy. Ann Intern Med 99: 127
Gralla RJ, Itri LM, Pisko SE, Squillante AE, Kelsen DP, Braun DW, Bordin LA, Braun TJ, Young CW (1981) Antiemetic efficacy of high-dose metoclopramide: Randomized trials with placebo and prochlorperazine in patients with chemotherapy-induced nausea and vomiting. N Engl J Med 305: 905–909
McDermed JE, Cohen JL, Joseph C, Strum SB (1985) Clinical pharmacokinetics of high-dose metoclopramide in cancer patients receiving cisplatin therapy. J Clin Oncol 3: 1400–1408
Ross-Lee LM, Eadie MJ, Hooper WD, Bochner F (1981) Single dose pharmacokinetics of metoclopramide. Eur J Clin Pharmacol 20: 465–471
Schuppan VD, Schmidt I, Heller M (1979) Untersuchungen zur Pharmakokinetik von Metoclopramid am Menschen. Arzneimittelforsch/Drug Res 29: 151–154
Taylor WB, Proctor SJ, Bateman DN (1984) Pharmacokinetics and efficacy of high-dose metoclopramide given by continuous infusion for the control of cytotoxic drug-induced vomiting. Br J Clin Pharmacol 18: 679–684
Tyson LB, Gralla RJ, Clark RA, Kris MG, Bosl GJ, Reich LM, Young CW (1984) High dose oral metoclopramide: dose-finding, efficacy and preliminary pharmacokinetic evaluation. Proc Am Soc Clin Oncol 3: 102
Wright GJ, Pitts JE (1984) Bioavailability of metoclopramide from tablet and liquid dosage forms relative to intravenous injection. Clin Pharmacol Ther 35: 284
Wright GJ, Melikian AP, Crowe JT (1984) Dose proportionality of metoclopramide. Clin Pharmacol Ther 35: 284
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Taylor, W.B., Bateman, D.N. Oral bioavailability of high-dose metoclopramide. Eur J Clin Pharmacol 31, 41–44 (1986). https://doi.org/10.1007/BF00870983
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00870983