Summary
In an open trial, the pharmacokinetics of ramipril and its active metabolite ramiprilat were studied in 25 hypertensive patients with various degrees of renal insufficiency given 5 mg ramipril p.o. for 14 days.
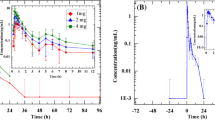
Ramipril was rapidly absorbed and reached a peak concentration after 1–2 h. Cmax was greater in patients with severe renal insufficiency, which might indicate a reduced renal elimination rate, although, the rapid decline of the concentration-time curve for ramipril was almost independent of renal function. The mean initial apparent half-lives on Days 1 and 12, respectively, were 2.8 and 3.4 h (Group I: creatinine clearance 5–15 ml/min), 1.8 and 2.3 h (Group II: creatinine clearance 15–40 ml/min), and 1.9 and 1.9 h (Group III: creatinine clearance 40–80 ml/min). No accumulation was observed after multiple dosing.
In contrast, the kinetics of its active acid metabolite ramiprilat was significantly influenced by renal function. The mean times to the peak plasma concentration were 5.7 h in Group I, 4.4 h in Group II and 3.8 h in Group III. The initial decline in plasma ramiprilat was dependent upon renal function; the mean initial apparent half-lives (Days 1 and 12, respectively) were 16.0 and 14.8 h (Group I), 10.1 and 9.5 h (Group II) and 10.6 and 8.0 h (Group III). Mean trough concentrations and absolute accumulation also increased with worsening renal function, and the renal clearance of ramiprilat was significantly correlated with the creatinine clearance. The subsequent long terminal phase at low plasma ramiprilat concentrations represented slow dissociation of the ACE-inhibitor complex.
The study indicates that in patients with severe renal insufficiency (creatinine clearance below 30 ml/min) smaller doses of ramipril are required than in patients with normal or borderline renal function.
Similar content being viewed by others
References
Becker RHA, Schölkens BA, Unger T, Linz W (1987) Ramipril: Review of pharmacology. Am J Cardiol 59: 3D-11D
Cushman DW, Cheung HS, Sabo EF, Ondetti MA (1977) Design of a potent competitive inhibitor of angiotensin converting enzyme. Biochemistry 16: 5484–5491
Atkinson AR, Morton JJ, Brown JJ, Davies DL, Fraser R, Kelly P, Lever AF, Lechie B, Robertson JIS (1980) Captopril in clinical hypertension: Changes in components of the renin angiotensin system and in body composition in relation to fall in blood pressure with a note on measurement of angiotensin II during converting enzyme inhibition. Br Heart J 44: 290–296
Dzau VJ, Colucci WS, Williams GH, Curfman G, Meggs L, Hollenburg NK (1980) Sustained effectiveness of converting enzyme inhibition in patients with severe congestive heart failure. N Engl J Med 302: 1373–1379
Sharpe DN, Murphy J, Coxon R, Hannan SF (1984) Enalapril in patients with chronic heart failure: A placebo-controlled, randomized double-blind study. Circulation 70: 271
Gavras H, Biollaz J, Waeber B, Brunner HR, Gavras I, Davies RO (1981) Antihypertensive effect of the new oral angiotensin converting enzyme inhibitor “MK-421”. Lancet 2: 543–547
Ferguson RR, Vlasses PH, Swanson BN (1982) Effects of enalapril, a new converting enzyme inhibitor, in hypertension. Clin Pharmacol Ther 32: 48–53
de Leeuw PW, Birkenhäger WH (1987) Short- and long-term effects of ramipril in hypertension. Am J Cardiol 59: 79D-82D
Crozier IG, Ikram H, Nicholls MG, Jans S (1987) Acute hemodynamic, hormonal and electrolyte effects of ramipril in severe congestive heart failure. Am J Cardiol 59: 155D-164D
Eckert H-G, Badian MJ, Gantz D, Kellner H-M, Volz M (1984) Pharmacokinetics and biotransformation of HOE 498 in rat, dog and man. Arzneimittelforsch 34: 1435–1447
Witte PU, Irmisch R, Hajdù P, Metzger H (1984) Pharmacokinetics and pharmacodynamics of a novel orally active angiotensin converting enzyme inhibitor (HOE 498) in healthy subjects. Eur J Clin Pharmacol 27: 577–581
Aurell M, Delin K, Herlitz H, Ljungman S, Witte PU, Irmisch R (1987) Pharmacokinetics and pharmacodynamics of ramipril in renal failure. Am J Cardiol 59: 65D-69D
Debusmann ER, Ocón Pujadas J, Lahn W, Irmisch R, Jané F, Shing Kuan T, Mora J, Walter U, Eckert H-G, Hajdù P, Metzger H (1987) Influence of renal function on pharmacokinetics of ramipril (HOE 498). Am J Cardiol 59: 70D-78D
Bartels H, Böhmer M, Heierle C (1972) Serum Kreatininbestimmung ohne Enteiweißen. Clin Chim Acta 37: 193–197
Eckert H-G, Münscher G, Oekonomopulos R, Strecker H, Urbach H, Wissmann H (1985) A radioimmunoassay for the angiotensin converting enzyme inhibitor ramipril and its active metabolite. Arzneimittelforsch 35: 1251–1256
Schmidt D, Keller A (1985) Eine empfindliche Methode zur Bestimmung des ACE-Hemmers HOE 498 und dessen Metaboliten in Humanurin mittels Capillarsäulen-Gas-Chromatographie. Fresenius Z Anal Chem 320: 731
SAS User's Guide 1982. SAS User's Guide: Statistics; Version 5th edn, 1985
Rommel AJ, Pierides AM, Heald A (1980) Captopril elimination in chronic renal failure. Clin Pharmacol Ther 30: 282
Duchin KL, Singhvi SM, Willard DA, Migdalof BH, McKinstry DN (1982) Captopril kinetics. Clin Pharmacol Ther 31: 452–458
Till AE, Gomez HJ, Hichens M, Bolognese JA, McNabb WR, Brooks BA, Noormohamed F, Lant AF (1984) Pharmacokinetics of repeated single doses of enalapril maleate (MK-421) in normal volunteers. Biopharm Drug Dispos 5: 273–280
Kubo SH, Cody RJ (1985) Clinical pharmacokinetics of the angiotensin converting enzyme inhibitors. Clin Pharmacokinet 10: 377–391
Lowenthal DT, Irvin JD, Merill D, Saris S, Ulm E, Goldstein S, Hichens M, Klein L, Till A, Harris K (1985) The effect of renal function on enalapril kinetics. Clin Pharmacol Ther 38: 661–666
Kelly JG, Doyle G, Donohue J, Laher M, Vandenburg MJ, Currie WJC, Cooper WD (1986) Pharmacokinetics of enalapril in normal subjects and patients with renal impairment. Br J Clin Pharmacol 21: 63–69
Fruncillo RJ, Rocci MJ, Vlasses PH, Mojaverian P, Shepley K, Clementi RA, Oren A, Smith RD, Till AE, Riley LJ, Krishna G, Narins RG, Ferguson RK (1987) Disposition of enalapril and enalaprilat in renal insufficiency. Kidney Int 31: (Suppl 20) 117S-122S
Duchin KL, McKinstry DN, Cohen AI, Migdalof BH (1988) Pharmacokinetics of captopril in healthy subjects and in patients with cardiovascular diseases. Clin Pharmacokinet 14: 241–259
Ulm EH, Hichens M, Gomez HJ, Till AE, Hand E, Vassil TC, Biollaz J, Brunner HR, Schelling JL (1982) Enalapril maleate and a lysine analogue (MK-521): Disposition in man. Br J Clin Pharmacol 14: 357–362
Ulm EH (1983) Enalapril maleate (MK-421), a potent, non-sulfhydryl angiotensin-converting enzyme inhibitor. Drug Metab Rev 14: 99–110
Singhvi SM, Duchin KL, Willard DA, McKinstry DN, Migdalof BH (1982) Renal handling of captopril: Effect of probenecid. Clin Pharmacol Ther 32,2: 182–189
Author information
Authors and Affiliations
Additional information
Abbreviated poster presentations of these results were given to the 12th Scientific Meeting of the International Society of Hypertension (May 22–26, 1988, Kyoto, Japan), and at the satellite symposium: “Antihypertensive Strategy using Angiotensin Converting Enzyme Inhibitors”. The poster will be published as a short paper in a supplement to J Cardiovasc Pharmacol
Rights and permissions
About this article
Cite this article
Schunkert, H., Kindler, J., Gassmann, M. et al. Pharmacokinetics of ramipril in hypertensive patients with renal insufficiency. Eur J Clin Pharmacol 37, 249–256 (1989). https://doi.org/10.1007/BF00679779
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00679779