Summary.
The pharmacokinetics of oral and i. v. propafenone and its major metabolites have been investigated in 8 healthy subjects.
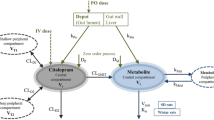
The total body clearance of propafenone was 963 ml/min, the terminal half-life 198 min and its absolute bioavailability was 15.5%. The two active metabolites (5-hydroxypropafenone and N-depropylpropafenone) showed non-linear kinetics in that both the dose-corrected area under the serum concentration-time curve and the amount excreted in the urine were larger after oral dosing. This resulted in considerably higher serum concentrations of the metabolites despite comparable serum concentrations of the parent compound. Thus, the concentration-effect relationship in the same patient may differ after oral and intravenous doses if concentrations of the active metabolite(s) are not taken into consideration.
Although the mechanism of the nonlinearity is not clear, the data indicate that it may be due to saturable biliary excretion of the metabolites.
Similar content being viewed by others
References
Connolly SJ, Kates RE, Lebsack CS, Echt DS, Mason JW, Winkle RA (1983) Clinical efficacy and electrophysiology of oral propafenone for ventricular tachycardia. Am J Cardiol 52: 1208–1213
Coumel P, Leclercq J-F, Assayag P (1984) European experience with the antiarrhythmic efficacy of propafenone for supraventricular and ventricular arrhythmias. Am J Cardiol 54: 60D-66D
Salerno DM, Granrud G, Sharkey P, Asinger R, Hodges M (1984) A controlled trial of propafenone for treatment of frequent and repetitive ventricular premature complexes. Am J Cardiol 53: 77–83
Hollmann M, Brode E, Hotz D, Kaumeier S, Kehrhahn OH (1983) Investigations on the pharmacokinetics of propafenone in man. Drug Res 33: 763–770
Connolly S, Lebsack C, Winkle RA, Harrison DC, Kates RE (1984) Propafenone disposition kinetics in cardiac arrhythmia. Clin Pharmacol Ther 36: 163–168
Kates RE, Yee YG, Winkle RA (1985) Metabolic cumulation during chronic propafenone dosing in arrhythmia. Clin Pharmacol Ther 37: 610–614
Siddoway LA, Thompson KA, McAllister CB, Wang T, Wilkinson GR, Roden DM, Woosley LR (1987) Polymorphism of propafenone metabolism and disposition in man: Clinical and pharmacokinetic consequences. Circulation 75: 785–791
von Philipsborn G, Gries J, Hofmann HP, Kreiskott H, Kretzschmar R, Muller CD, Raschack M, Teschendorf HJ (1984) Pharmacological studies on propafenone and its main metabolite 5-hydroxypropafenone. Drug Res 34: 1489–1497
Haefeli W, Vozeh S, Ha HR, Oti K, Follath F (1988) Pharmacologic activity of 5-hydroxypropafenone, a major metabolite of propafenone in man. Clin Res 36: 364A
Gibaldi M, Perrier D (1982) Pharmacokinetics Vol 15. Dekker, New York
Pang KS (1985) A review of metabolite kinetics. J Pharmacokinet Biopharm 13: 633–662
Dixon WJ, Mood AM (1946) The statistical sign test. J Am Stat Assoc 41: 557–566
Giani P, Landolina M, Giudici V, Bianchini C, Ferrario G, Marchi S, Riva E, Latini R (1988) Pharmacokinetics and pharmacodynamics of propafenone during acute and chronic administration. Eur J Clin Pharmacol 34: 187–194
Hege HG, Hollmann M, Kaumeier S, Lietz H (1984) The metabolic fate of2H-labelled propafenone in man. Eur J Drug Metabol Pharmacokinet 9: 41–55
Verbeeck RK, Branch RA, Wilkinson GR (1981) Drug metabolites in renal failure: Pharmacokinetic and clinical implications. Clin Pharmacokinet 6: 329–345
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Vozeh, S., Haefeli, W., Ha, H.R. et al. Nonlinear kinetics of propafenone metabolites in healthy man. Eur J Clin Pharmacol 38, 509–513 (1990). https://doi.org/10.1007/BF02336693
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02336693