Summary
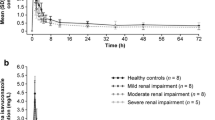
The pharmacokinetics and pharmacodynamics of the ACE inhibitor quinaprilat have been studied in six chronic haemodialysis (HD) patients and in six patients undergoing continuous ambulatory peritoneal dialysis (CAPD) after a single oral dose of 2.5 mg quinapril. Mean tmax and Cmax values (SEM) for quinaprilat in interdialytic HD patients were 4.0 (0) h and 84 (8.4) ng · ml−1 respectively, and they did not differ significantly from those in CAPD patients (4.7 (0.7) h and 64 (5.7) ng · ml−1). Elimination half lives were 30 (10.1) h (HD) and 34 (7.3) h (CAPD). Cmax, tmax, t/12, and AUC were increased and CL was decreased compared to data reported previously after giving 2.5 mg to healthy subjects. Peritoneal clearance was calculated as 0.1 (0.1) ml · min−1, thus less than 0.5 % of the dose were removed within 24 h by CAPD.
ACE activity was suppressed by more than 93 % between 4 and 24 h postdose (P < 0.001). It decreased in both groups with increasing plasma quinaprilat levels. Angiotensin II concentration compared to baseline was significantly decreased at 4 hours (−30.4±10 %) and 24 h (−30±9.9 %) (P < 0.05,n=11), while active plasma renin concentration was still significantly increased at 48 h postdose (+60.2±14.5 %,P < 0.01). Mean arterial pressure 24 h postdose was significantly (P < 0.05) decreased in HD (−12 mm Hg) and CAPD patients (− 20 mm Hg). Only two patients reported unwanted effects (fatigue, dizziness, nausea, and weakness).
In conclusion, due to its long lasting effect on ACE activity and on blood pressure in terminal renal failure a starting dose of quinapril 2.5 mg o. d. may be used in hypertensive HD and CAPD patients.
Similar content being viewed by others
References
Olson SC, Horvath AM, Michniewicz BM, Sedman AJ, Colburn WA, Welling PG (1989) The clinical pharmacokinetics of quinapril. Angiology 40: 351–359
Begg EJ, Robson RA, Bailey RR, Lynn KL, Frank GJ, Olson SC (1990) The pharmacokinetics and pharmacodynamics of quinapril and quinaprilat in renal impairment. Br J Clin Pharmacol 30: 213–220
Blum RA, Olson SC, Kohli RK, Horvath AM, Sedman AJ, Posvar EL (1990) Pharmacokinetics of quinapril and its active metabolite quinaprilat in patients on chronic hemodialysis. J Clin Pharmacol 30:938–942
Swartz RD, Starmann B, Horvath AM, Olson SC, Posvar EL (1990) Pharmacokinetics of quinapril and its active metabolite quinaprilat during continuous ambulatory, peritoneal dialysis. J Clin Pharmacol 30:1136–1141
Drummer OH, Workman BS, Miach PJ, Jarrott B, Louis WJ (1987) The pharmacokinetics of captopril and capropril disulfide conjugates in uraemic patients on maintenance dialysis comparison with patients with normal renal function. Eur J Clin Pharmacol 32:257–271
Fujimura A, Kajiyama J, Ebihara A, Iwashita K, Nomura Y, Kawahara Y (1986) Pharmacokineties and pharmacodynamics of captopril in patients undergoing continuous ambulatory peritoneal dialysis. Nephron 44:324–328
Fruncillo RJ, Rocci ML, Vlasses PH, Mojaverian P, Shepley K, Clementi RA, Oren A, Smith RD, Till AE, Riley LJ Jr., Krishna G, Narins RG, Ferguson RK (1987) Disposition of enalapril and enalaprilat in renal insufficiency. Kidney Int 31 [Suppl 20]: 117–122
Sedman AJ, Posvar E (1989) Clinical pharmacology of quinapril in healthy volunteers and in patients with hypertension and congestive heart failure. Angiology 40: 360–369
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Wolter, K., Fritschka, E. Pharmacokinetics and pharmacodynamics of quinaprilat after low dose quinapril in patients with terminal renal failure. Eur J Clin Pharmacol 44 (Suppl 1), S53–S56 (1993). https://doi.org/10.1007/BF01428395
Issue Date:
DOI: https://doi.org/10.1007/BF01428395