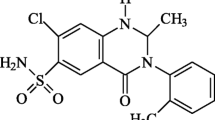
Abstract
Objectives: The present study was conducted to compare pharmacokinetic behaviors of nicardipine enantiomers given in different doses with different formulations of racemic nicardipine in healthy volunteers.
Methods: One or two 20-mg racemic nicardipine tablets, and a 40-mg sustained-release capsule of nicardipine were administered to eight healthy volunteers in a cross-over fashion and pharmacokinetic parameters were evaluated. Enantiomer concentrations were determined by GC-MS combined with chiral stationary phase HPLC.
Results and conclusions: Serum concentration of (+)-nicardipine was approximately 2–3 times higher than that of (−)-nicardipine in 20- and 40-mg doses of conventional formulations and a non-linear increase in bioavailability with dose was demonstrated. The value for AUC of (+)-nicardipine was approximately 2.3–2.8 times greater than that of the (−)-nicardipine (P < 0.05) when 20 and 40 mg racemic nicardipine were administered in a conventional preparation. Relative bioavailability of the sustained-release preparation vs the conventional preparation was 28% and 44% for (+)- and (−)-nicardipine, respectively, for the 40-mg dose.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received: 23 September 1996 / Accepted in revised form: 23 February 1997
Rights and permissions
About this article
Cite this article
Inotsume, N., Iwaoka, T., Honda, M. et al. Pharmacokinetics of nicardipine enantiomers in healthy young volunteers. E J Clin Pharmacol 52, 289–292 (1997). https://doi.org/10.1007/s002280050292
Issue Date:
DOI: https://doi.org/10.1007/s002280050292