Abstract
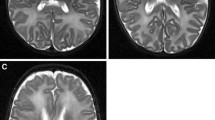
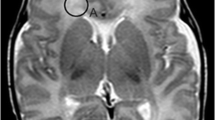
Our purpose was to study the magnetic resonance (MR) signal intensity of the perirolandic gyri perinatally and to correlate it with the histological findings in formalinfixed brains, focusing on myelination. MRI of 20 neurologically normal neonates and infants, of 37–64 weeks postconception (PCA), were studied retrospectively. We reviewed four formalin-fixed brains of infants 37–46 weeks PCA microscopically. The posterior cortex of the precentral gyrus (P-PRE) and the anterior cortex of the postcentral gyrus (A-PST) had different signal intensity from the adjacent surrounding cortex. On T1-weighted images P-PRE and A-PST gave higher signal 41–44 weeks PCA; on T2-weighted images, they gave lower signal 37–51 weeks PCA. Histological examination revealed very little myelination of the nerve fibres within both the P-PRE and the A-PST, while considerable myelination was present in the internal capsule and central corona radiata. The changes in signal intensity in the perirolandic gyri may reflect not only the degree of myelination but also the more advanced development of the nerve cells, associated with rapid proliferation and formation of oligodendroglial cells, synapses and dendrites. They could be another important landmark for brain maturation.
Similar content being viewed by others
References
Johnson MA, Pennock JM, Bydder GM, et al (1983) Clinical NMR imaging of the brain in children: normal and neurologic disease. AJR 141:1005–1018
Pennock JM, Bydder GM, Dubowitz LMS, Johnson MA (1986) Magnetic resonance imaging of the brain in children. Magn Reson Imaging 4:1–9
Dubowitz LMS, Pennock JM, Johnson MA, Bydder GM (1986) High resolution magnetic imaging of the brain in children. Clin Radiol 37:113–117
Holland BA, Haas DK, Norman D, Brant-Zawadzki M, Newton TH (1986) MRI of normal brain maturation. AJNR 7:201–208
McArdle CB, Richardson CJ, Nicholas DA, Mirfakhraee M, Hayden CK, Amparo EG (1987) Developmental features of the neonatal brain: MR imaging. I. Gray-white matter differentiation and myelination. Radiology 162:223–229
Nowell MA, Hackney DB, Zimmermann RA, Bilaniuk LT, Grossman RI, Goldberg HI (1987) Immature brain: spin-echo pulse sequence parameters for high contrast MR imaging. Radiology 167:272–273
Barkovich AJ, Kjos BO, Jackson DE Jr, Norman D (1988) Normal maturation of neonatal and infant brain: MR imaging at 1.5T. Radiology 166:173–180
Martin E, Kikinis R, Zuerrer M, Boesch C, Briner J, Kewitz G, Kaelin P (1988) Developmental stages of human brain: an MR study. J Comput Assist Tomogr 12:917–922
Martin E, Boesch C, Grutter R, et al (1988) Magnetic resonance in pediatric research and clinical diagnosis. II. Investigation of the developing brain in neonates, infants and small children. Helv Paediatr Acta 43:76–86
Dietrich RB, Bradley WG, Zaragona EJIV, et al (1988) MR evaluation of early myelination patterns in normal and developmentally delayed infants. AJNR 9:69–76
Baierl P, Forster C, Fendel H, Naegele M, Fink U, Kenn W (1988) Magnetic resonance imaging of normal and pathological white matter maturation. Pediatr Radiol 18:183–9
Hayakawa K, Konishi Y, Kuriyama M, Konishi K, Matsuda T (1990) Normal brain maturation in MRI. Eur J Radiol 12:208–215
Barkovich AJ (1990) Pediatric neuroimaging. Raven Press, New York, pp 5–34
Gilles HH (1991) Perinatal neuropathology. In: Davis RL, Robertson DM (eds) Textbook of neuropathology. Williams & Wilkins, Baltimore, pp 290–330
Yakovlev PI, Lecours AR (1967) The myelogenetic cycles of regional maturation of the brain. In: Minkowski A (ed) Regional development of the brain in early life. Blackwell, Oxford, pp 3–70
Brody BA, Kinney HC, Kloman AS, Gilles FH (1987) Sequence of central nervous system myelination in human infancy. I. An autopsy study of myelination. J Neuropathol Exp Neurol 46: 283–301
Kinney HC, Brody BA, Kloman AS, Gilles FH (1988) Sequence of central nervous system myelination in human infancy. II. Patterns of myelination in an autopsied infants. J Neuropathol Exp Neurol 47:217–234
Conel JL (1941) The postnatal development of the human cerebral cortex, vol II. The cortex of the one-month infant. Harvard University Press, Cambridge
Conel JL (1947) The postnatal development of the human cerebral cortex, vol III. The cortex of the three-month infant. Harvard University Press, Cambridge
Conel JL (1951) The postnatal development of the human cerebral cortex, vol IV. The cortex of the six-month infant. Harvard University Press, Cambridge
Dobbing J, Sands J (1973) Quantitative growth and development of human brain. Arch Dis Child 48:757–767
Davison AN, Dobbing J (1968) The developing brain. In: Davison AN, Dobbing J (eds) Applied neurochemistry. Blackwell, Oxford, p 253
Huttenlocher PR, Courten C de, Garey LJ, Van der Loos H (1982) Synaptogenesis in human visual cortex-evidence for synapse elimination during normal development. Neurosci Lett 33: 247–252
Becker L, Armstrong DL, Chan F, Wood MM (1984) Dendritic development in human occipital cortical neurons. Dev Brain Res 13:117–124
Takashima S, Chan F, Becker LE, Armstrong DL (1980) Morphology of the developing visual cortex of the human infant. A quantitative and qualitative Golgi study. J Neuropathol Exp Neurol 39:487–501
Aoki S, Okada Y, Nishimura K, Barkovich AJ, Brasch RC, Norman D (1989) Normal deposition of brain iron in childfood and adolescence: MR imaging at 1.5 T. Radiology 172:381–385
Höck A, Demmel H, Schicha K, Kasperek K, Feinendegen LE (1975) Trace element concentration in human brain. Brain 98:49–64
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Korogi, Y., Takahashi, M., Sumi, M. et al. MR signal intensity of the perirolandic cortex in the neonate and infant. Neuroradiology 38, 578–584 (1996). https://doi.org/10.1007/BF00626104
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00626104