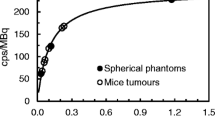
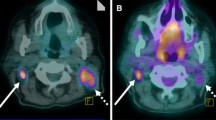
Abstract
We present a method to assess quantitatively the immunological characteristics of tumours using radiolabelled monoclonal antibody and positron emission tomography (PET) to improve dosimetry for radioimunotherapy. This method is illustrated with a glioma patient who was injected with 96.2 MBq of iodine-124 labelled 3F8, a murine antibody (IgG3) specific against the ganglioside GD2. Serial PET scans and plasma samples were taken over 11 days. A three-compartment model was used to estimate the plasma to tumour transfer constant (K 1), the tumour to plasma transfer constant k 2, the association and dissociation constants (k 3, k 4) of antibody binding, and the binding potential. Tumour radioactivity peaked at 18 h at 0.0045% ID/g. The kinetic parameters were estimated to be: K 1 = 0.048 ml h−1 g−1, k 2 = 0.16 h−1, k 3 = 0.03 h−1, k 4 = 0.015 h−1 and BP = 2.25. Based on these kinetic parameters, the amount of tumour-bound radiolabelled monoclonal antibody was calculated. This method permits estimates of both macrodosimetry and microdosimetry at the cellular level based on in vivo non-invasive measurement.
Similar content being viewed by others
References
Pentlow KS, Graham MC, Lambrecht RM, Cheung N-KV, Larson SM. Quantitative imaging of I-124 using positron emission tomography with applications to radioimmunodiagnosis and radioimmunotherapy. Med Phys 1991; 18:357–366
Miraldi F. Monoclonal antibodies and neuroblastoma. Semin Nucl Med 1989; 19:282–294
Rubin SC, Kairemo KJA, Brownell AL, et al. High-resolution positron emission tomography of human ovarian cancer in nude rats using I-124 labelled monoclonal antibodies. Gynecol Oncol 1993; 48:61–67
Larson SM, Pentlow KS, Volkow ND, et al. PET scanning of I-124 3F8 as an approach to tumor dosimetry during treatment planning for radioimmunotherapy in a child with neuroblastoma. J Nucl Med 1992; 33:2020–2023
Wilson CB, Snook DE, Dhokia B, et al. Quantitative measurement of monoclonal antibody distribution and blood flow using positron emission tomography and 124 iodine in patients with breast cancer. Int J Cancer 1991; 47:344–347
Cheung N-KV, Saarinen UM, Neely JE, Landmeier B, Donovan D, Coccia P. Monoclonal antibodies to a glycolipid antigen on human neuroblastoma cells. Cancer Res 1985; 45:2642–2649
Bosslet K, Mennel HD, Rodden F, Bauer BL, Wagner F, Altmannsberger A, Sedlack HH, Wiegandt H. Monoclonal antibodies against epitopes on ganglioside GD2 and its lactones. Markers for gliomas and neuroblastomas. Cancer Immunol Immunother 1989; 29:171–178
Finn R, Cheung N-KV, Divgi C, St. Germain J, Graham MC, Pentlow KS, Larson SM. Technical challenges associated with the radiolabeling of monoclonal antibodies utilizing short-lived, positron emitting radionuclides. Nucl Med Biol 1991; 18:9–13
Blasberg RG, Nakagawa H, Bourdon MA, Groothuis DR, Patlak CS, Bigner DD. Regional localization of a glioma-associated antigen defined by monoclonal antibody 81C6 in vivo: kinetics and implications for diagnosis and therapy. Cancer Res 1987; 47:4432–4443
Hawkins RA, Phelps ME, Huang SC. Effects of temporal sampling, glucose metabolic rates, and disruptions of the blood-brain barrier on the FDG model with and without a vascular compartment: studies in human brain tumors with PET. J Cereb Blood Flow Metab 1986; 6:170–183
MIRD. J Nucl Med SuppI 1, Feb 1968
Kassis AI, Adelstein SJ, Haydock C, Sastry KSR. Radiotoxicity of Se-75 and S-35: theory and application to a cellular model. Radiat Res 1980; 84:407–425
Cole A. Absorption of 20-eV to 50000-eV electron beams in air and plastic. Radiat Res 1969; 38:7–33
Mintun MA, Raichle ME, Kilbourn MR, Wooten GF, Welsh MJ. Quantitative model for the in vivo assessment of drug binding sites with positron emission tomography. Ann Neurol 1984; 15:217–227
Browne E, Firestone RB, Shirley VS, eds. Table of radioactive isotopes. New York: John Wiley, 1986
Russell DS, Rubinstein LJ. Pathology of tumours of the nervous system. Williams & Wilkins: Baltimore, 1989
Epenetos AA, Courtenary-Luck N, Pickering D, et al. Antibody guided irradiation of brain glioma by erterial infusion of radioactive monoclonal antibody against epidermal growth factor receptor and blood group A antigen. Br J Med 1985; 290:1463–1466
Kaofonos HP, Pawlikowska TR, Hemingway A, et al. Antibody guided diagnosis and therapy of brain gliomas using radiolabelled monoclonal antibodies against epidermal growth factor receptor and placental alkaline phosphatase. J Nucl Med 1989; 30:1636–1645
Bergh J, Nilsson S, Liljedahl C, et al. Radioimaging of human malignant gliomas using indium-labelled monoclonal anti bodies. Nucl Med Commun 1990; 11:437–444
Zalutsky MR, Moseley RP, Coakham HB, Coleman RE, Bigner DD. Pharmacokinetics and tumor localization of iodine-131 labelled anti-tenascin monoclonal antibody 81C6 in patients with gliomas and other intracranial malignancies. Cancer Res 1989 49:2807–2813
Bosslet K, Mennel HD, Rodden F, et al. In: Oettgen HF, ed. Gangliosides and cancer. Weinheim: Verlag Chemie; 1989: 317
Miraldi FD, Nelson AD, Kraly C, et al. Diagnostic imaging of human neuroblastoma with radiolabeled antibody. Radiology 1986; 161:413–418
Yeh SDJ, Larson SM, Burch, et al. Radioimmunodetection of neuroblastoma with iodine-131–3178: correlation with biopsy, iodine-131-metaiodobenzylguanidine and standard diagnostic, modalities. J Nucl Med 1991; 32:769–776
Cheung N-KV, Yeh SDJ, Gulati S, Burch L, Kushner BH, Larson SM. Iodine-131–3F8 targeted radiotherapy of neuroblastoma (NB): a phase I clinical trial. Proc AACR 1990; 31:284
Waldmann TA. Metabolism of immunoglobulins. Prog Allergy 1969; 13:1–110
Waldmann TA. Methods for the study of the metabolism of immunoglobulins. Methods Enzymol 1985; 116:210–214
Yokoyama K, Carrasquillo JA, Chang AE, et al. Differences in biodistribution of indium-111 and iodine-131 labeled B72.3 monoclonal antibodies in patients with colorectal cancer. J Nucl Med 1989; 30:320–327
Larson SM, Brown JP, Wright PW, et al. Imaging of melanoma with I-131 labelled monoclonal antibodies. J Nucl Med 1983; 24:123–129
Lotze MT, Carrasquillo JA, Weinstein JN, et al. Monoclonal antibody imaging of human melanoma; radioimmunodetection, by subcutaneous or systemic injection. Ann Surg 1986; 204:19–31
Makrigiorgos GM, Adelstein SJ, Kassis AI. Limitations of conventional internal dosimetry at the cellular level. J Nucl Med 1989; 30:1856–1864
Kassis AI, Sastry KSR, Adelstein SJ. Kinetics of uptake, retention, and radiotoxicity of 125IUDR in mammalian cells: implications of localized energy deposition by Auger processes. Radiat Res 1987; 109:78–89
Kassis AI, Makrigiorgos GM, Adelstein SJ. Dosimeric considerations and therapeutic potential of Auger electron emitters. In: Adelstein SJ, ed. Dosimetry of administered radionuclides. Proceedings of a Conference, Washington D.C. 1989, American College of Nuclear Physicians (publication # 90-2), pp 257–274
Author information
Authors and Affiliations
Additional information
Correspondence to: F. Daghighian
Rights and permissions
About this article
Cite this article
Daghighian, F., Pentlow, K.S., Larson, S.M. et al. Development of a method to measure kinetics of radiolabelled monoclonal antibody in human tumour with applications to microdosimetry: positron emission tomography studies of iodine-124 labelled 3F8 monoclonal antibody in glioma. Eur J Nucl Med 20, 402–409 (1993). https://doi.org/10.1007/BF00208998
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00208998