Abstract
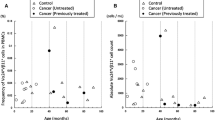
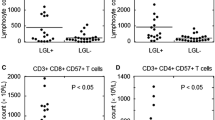
The effect of subcutaneous recombinant interleukin-2 (rIL-2) therapy on the “activation status” of peripheral blood lymphocytes (PBL) of 17 renal cell carcinoma patients was investigated in a longitudinal study. The expression of the activation markers HLA-Dr and CD25 on cytotoxic T cells, helper T cells, and natural killer (NK) cells, was analysed using two-colour flow cytometry of whole-blood samples. In addition, the ability of isolated PBL to proliferate in vitro in response to various stimuli was investigated. The absolute amounts of NK cells and HLA-DR-expressing NK cells increased continuously during the whole course of therapy. The absolute amounts of T cells and HLA-Dr-expressing T cells, however, showed an early increase only during the first 1 or 2 weeks of therapy, after which the absolute amounts of HLA-Dr-expressing T cells decreased. In particular, the absolute amount of HLA-Dr-expressing CD8bright+ T cells was significantly lowered in the second half of therapy. PBL collected on day 7 of therapy (post-cycle-1 PBL) showed, as compared to those collected prior to therapy (pretherapy PBL), a decreased proliferative response in vitro after stimulation with phytohaemagglutinin, concanavalin A, soluble CD3 mAb (WT32) or rIL-2. This decreased in vitro response of post-cycle-1 PBL was also reflected in a decrease in the percentage of CD8bright+ T cells expressing HLA-Dr in cultures with rIL-2 or CD3 mAb, in contrast to cultures of pretherapy PBL, which showed an increase of this percentage. We conclude that T cells are the predominantly stimulated subpopulation during the first 2 weeks of subcutaneous rIL-2 therapy. The significant decrease in the absolute amounts of HLA-Dr-expressing T cells in the peripheral blood during the second half of therapy may partly be explained by a decreased responsiveness to rIL-2, but a selective redistribution of HLA-Dr-expressing cells may also be involved.
Similar content being viewed by others
References
Cantrell DA, Smith KA (1984) The interleukin-2 T-cell system: a new cell growth model. Science 224: 1312
Ellis TM, Creekmore SP, McMannis JD, Braun DP, Harris JA, Fisher RI (1988) Appearance and phenotypic characterization of circulating leu 19+ cells in cancer patients receiving recombinant interleukin 2. Cancer Res 48: 6597
Favrot MC, Combaret V, Negrier S, Philip I, Thiesse P, Freydel C, Bijmann JT, Franks CR, Mercatello A, Phillip T (1990) Functional and immunophenotypic modifications induced by interleukin-2 did not predict response to therapy in patients with renal cell carcinoma. J Biol Response Mod 9: 167
Hank JA, Sosman JA, Kohler PC, Bechhofer R, Storer B, Sondel PM (1990) Depressed in vitro T cell responses concomitant with augmented interleukin-2 responses by lymphocytes from cancer patients following in vivo treatment with interleukin-2. J Biol Response Mod 9: 5
Hänninen EL, Körfer A, Hadam M, Schneekloth C, Dallmann I, Menzel T, Kirchner H, Poliwoda H, Atzpodien J (1991) Biological monitoring of low-dose interleukin 2 in humans: soluble interleukin 2 receptors, cytokines, and cell surface phenotypes. Cancer Res 50: 6312
Ko HS, Fu SM, Winchester RJ, Yu DTY, Kunkel HG (1979) Ia determinants on stimulated human T lymphocytes. Occurence on mitogen- and antigen-activated T cells. J Exp Med 150: 246
Rosenberg SA, Lotze MT, Muul, IM, Chang AE, Avis FP, Leitman S, Linehan M, Robertson CN, Lee RE, Rubin JT, Seipp CA, Simpson CG, White DE (1987) A progress report on the treatment of 157 patients with advanced cancer using lymphokine-activated killer cells and interleukin-2 or high-dose interleukin-2 alone. N Engl J Med 316: 889
Sleijfer DTh, Janssen RAJ, Willemse PHB, Martens A, de Leij L, de Vries EGE, Mulder NH (1990) Low-dose regimen of interleukin-2 for metastatic renal carcinoma. Lancet 335: 1522
Sleijfer DTh, Janssen RAJ, Buter J, de Vries EGE, Willemse PHB, Mulder NH (1992) Phase II study of subcutaneous interleukin-2 in unselected patients with advanced renal cell cancer on an outpatient basis. J Clin Oncol 10: 1119
Sondel PM, Kohler PC, Hank JA, Moore KH, Rosenthal NS, Sosman JA, Bechhofer R, Storer B (1988) Clinical and immunological effects of recombinant interleukin 2 given by repetitive weekly cycles to patients with cancer. Cancer Res 48: 2561
Talmadge JE, Wiltrout RH, Counts DF, Herberman RB, McDonald T, Ortaldo JR (1986) Proliferation of human peripheral blood lymphocytes induced by recombinant human interleukin 2: contribution of large granular lymphocytes and T lymphocytes. Cell Immunol 102: 261
Tax WJM, Willems HW, Reekers PPM, Capel PJ, Koene RAP (1983) Polymorphism in mitogenic effect of IgG1 monoclonal antibodies against T3 antigen on human T cells. Nature 304: 445
Thi Bich-Thuy L, Lane HC, Fauci AS (1986) Recombinant interleukin-2 induced polyclonal proliferation of in vitro unstimulated human peripheral blood lymphocytes. Cell Immunol 98: 396
Urba WJ, Steis RG, Longo DL, Kopp WC, Maluish AE, Marcon L, Nelson DL, Stevenson HC, Clark JW (1990) Immunomodulatory properties and toxicity of interleukin 2 in patients with cancer. Cancer Res 50: 185
Weil-Hillman G, Hank JA, Rosenthal NS, Sondel PM (1988) Transient decrease in Il-2-responsive lymphocytes 24 hours after initiation of continuous IL-2 infusion in cancer patients. J Biol Response Mod 7: 424
Weil-Hillman G, Fisch P, Prieve AF, Sosman JA, Hank JA, Sondel PM (1989) Lymphokine-activated killer activity induced by in vivo interleukin 2 therapy: predominant role for lymphocytes with increased expression of CD2 and leu 19 antigens but negative expression of CD16 antigens. Cancer Res 49: 3680
Weil-Hillman G, Schell K, Segal DM, Hank JA, Sosman JA, Sondel PM (1991) Activation of human T cells obtained pre- and post interleukin-2 (IL-2) therapy by anti-CD3 monoclonal antibody plus IL-2: inplications for combined in vivo treatment. J Immunother 10: 276
West WH, Tauer KW, Yanelli JR, Marshall GD, Orr DW, Thjurman GB, Oldhan RK (1987) Constant infusion of recombinant interleukin-2 in adoptive immunotherapy of advanced cancer. N Engl J Med 316: 898
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Janssen, R.A.J., Buter, J., Straatsma, E. et al. HLA-Dr-expressing CD8bright cells are only temporarily present in the circulation during subcutaneous recombinant interleukin-2 therapy in renal cell carcinoma patients. Cancer Immunol Immunother 36, 198–204 (1993). https://doi.org/10.1007/BF01741092
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01741092